The ancient wisdom of Hippocrates, who declared nearly 2,500 years ago that "All disease begins in the gut," is increasingly being validated by modern scientific inquiry. Our gut microbiome, a complex ecosystem of trillions of microorganisms, plays a pivotal role in our overall health, influencing everything from digestion and nutrient absorption to immune function and even mental well-being. When we nourish these microbial inhabitants with a diet rich in whole plant foods, they reciprocate by producing beneficial compounds such as butyrate, a short-chain fatty acid crucial for gut health, derived from dietary fiber. Conversely, a diet lacking in these essential nutrients and high in certain animal products can lead to the production of detrimental compounds, most notably trimethylamine N-oxide (TMAO).
TMAO is a metabolite produced by gut bacteria from precursors found in foods like red meat, eggs, seafood, and dairy products. For years, TMAO was primarily associated with cardiovascular diseases, including heart disease and stroke. However, a growing body of research has revealed a far more pervasive and concerning link between TMAO and a wide array of chronic health conditions. This expanding understanding has prompted significant scientific interest in understanding the mechanisms by which TMAO exerts its negative effects and, crucially, whether interventions targeting the gut microbiome can mitigate these risks.
TMAO’s Expanding Association with Chronic Disease
The initial identification of TMAO’s link to cardiovascular health marked a significant turning point in understanding its biological impact. Studies have consistently demonstrated a correlation between elevated TMAO levels and an increased risk of atherosclerosis, hypertension, and thrombotic events. However, the scientific community has since uncovered associations between TMAO and a much broader spectrum of diseases, underscoring its systemic influence.
Cardiovascular Disease and Stroke: The link between TMAO and cardiovascular disease is well-established. Elevated TMAO levels have been shown to contribute to the development of atherosclerosis by promoting inflammation and oxidative stress within blood vessels. This can lead to plaque buildup, narrowing of arteries, and an increased risk of heart attacks and strokes. Furthermore, TMAO has been linked to higher blood pressure and an increased likelihood of clot formation, particularly in individuals with atrial fibrillation, a common heart rhythm disorder. Research indicates that individuals with higher TMAO levels not only experience more severe strokes but also face a fourfold increased risk of mortality following such an event.
Cancer Risk: Perhaps one of the most alarming recent discoveries is the association between TMAO and an increased risk of various cancers, which collectively represent the second leading cause of death in the United States. While the precise mechanisms are still under investigation, TMAO’s pro-inflammatory and pro-oxidative properties are believed to play a significant role. Chronic inflammation can create an environment conducive to cellular mutations and tumor growth. Additionally, TMAO may contribute to DNA damage and disrupt protein folding, processes fundamental to maintaining cellular integrity and preventing cancerous transformations.
Neurodegenerative Diseases: The reach of TMAO extends to the brain, a territory once thought to be relatively insulated. TMAO has been detected in human cerebrospinal fluid, the fluid that bathes the brain and spinal cord. Studies have revealed higher TMAO levels in individuals experiencing mild cognitive impairment and those diagnosed with Alzheimer’s disease dementia. In the brain, TMAO has been shown to induce neuronal senescence, a state of cellular aging and dysfunction. It also exacerbates oxidative stress, impairs mitochondrial function – the powerhouses of our cells – and inhibits mTOR signaling, a critical pathway involved in cell growth and metabolism. These detrimental effects collectively contribute to brain aging and cognitive decline, highlighting a potential pathway linking gut health to neurological health.
Metabolic and Respiratory Disorders: TMAO’s influence is also apparent in metabolic and respiratory conditions. Individuals with higher TMAO levels are approximately 50% more likely to develop diabetes, the seventh leading cause of death in the U.S. The exact mechanisms are still being elucidated, but TMAO may interfere with insulin sensitivity and glucose metabolism. In the realm of respiratory health, TMAO has been linked to poorer outcomes in patients with chronic obstructive pulmonary disease (COPD), the fourth leading killer. While it’s suspected that the increased mortality in COPD patients with high TMAO might be indirectly due to associated cardiovascular disease, the compound’s presence independently predicts fatal outcomes in pneumonia patients, even in the absence of pre-existing heart conditions.
Kidney Disease: As the ninth leading cause of death, kidney disease is another condition where TMAO plays a significant role. TMAO levels are strongly correlated with kidney function, and high levels are predictive of fatal outcomes in patients with kidney disease. A longitudinal study revealed a stark difference in survival rates: over a five-year period, more than half of chronic kidney disease patients with average or higher TMAO levels died, whereas among those in the lowest third of TMAO levels, nearly 90% remained alive. This suggests that TMAO may not only be a marker of kidney dysfunction but could also actively contribute to its progression.
Exploring Interventions: The Fecal Transplant Enigma
Given the pervasive negative health impacts of TMAO, the question naturally arises: can we effectively lower these harmful levels? The most direct approach would be to modify dietary intake of TMAO precursors. Choline and carnitine, abundant in meat, eggs, and dairy, are the primary dietary sources for TMAO production. However, these nutrients are widespread, and complete restriction may not be practical or advisable for all individuals. This limitation has led researchers to explore more radical interventions, including the concept of altering the gut microbiome itself.
The idea of "swapping gut flora" gained traction, particularly with the notion of fecal microbiota transplantation (FMT). This procedure involves transferring fecal matter from a healthy donor to a recipient to re-establish a balanced gut microbiome. In the context of TMAO, the hypothesis was that transplanting gut bacteria from individuals who consume a plant-based diet – and thus produce little TMAO – could confer protection to recipients.

The Scientific Investigation into Vegan Fecal Transplants
A pivotal study explored the efficacy of vegan fecal transplants in reducing TMAO levels. In a double-blind, randomized, controlled trial, participants received either fecal material from vegan donors or their own processed fecal matter. The administration route involved a nasogastric tube, a procedure that, while effective for delivery, may not be ideal for long-term patient comfort or adherence.
The initial findings from this study presented a complex picture. While the intention was to introduce a microbiome that would suppress TMAO production, the results were not as straightforward as anticipated. One significant factor identified was the baseline microbiome of the vegan donors themselves. Unlike in previous observational studies where long-term vegans demonstrated virtually no TMAO production even after consuming meat, the vegans recruited for this transplant study were found to be producing TMAO themselves. This suggests that the definition of "vegan microbiome" in this context might have been less distinct than previously assumed, or that the duration of veganism among the donors was insufficient to fully reshape their gut flora’s metabolic output.
Furthermore, the study observed that approximately two weeks after the transplant, there was no significant reduction in TMAO levels in the recipients. This outcome could be attributed to several factors. Firstly, the limited baseline microbiome differences between the donors and recipients might have hindered a substantial shift in the recipient’s gut environment. Secondly, and critically, the study protocol allowed participants to continue their omnivorous diets after the transplant. This decision, while aimed at isolating the effect of the fecal transplant, may have inadvertently counteracted any potential benefits. The researchers acknowledged that "limited baseline microbiome differences and continuation of an omnivorous diet" could be contributing factors to the lack of improvement.
The Fundamental Role of Diet: No Shortcuts to a Healthy Gut
The implications of these findings are profound. They suggest that simply altering the microbial composition of the gut may not be a panacea for TMAO-related health issues if the underlying dietary habits remain unchanged. The researchers deliberately avoided switching participants to a plant-based diet during the trial to isolate the effect of the fecal transplant. They recognized that dietary changes alone are potent modulators of the gut microbiome.
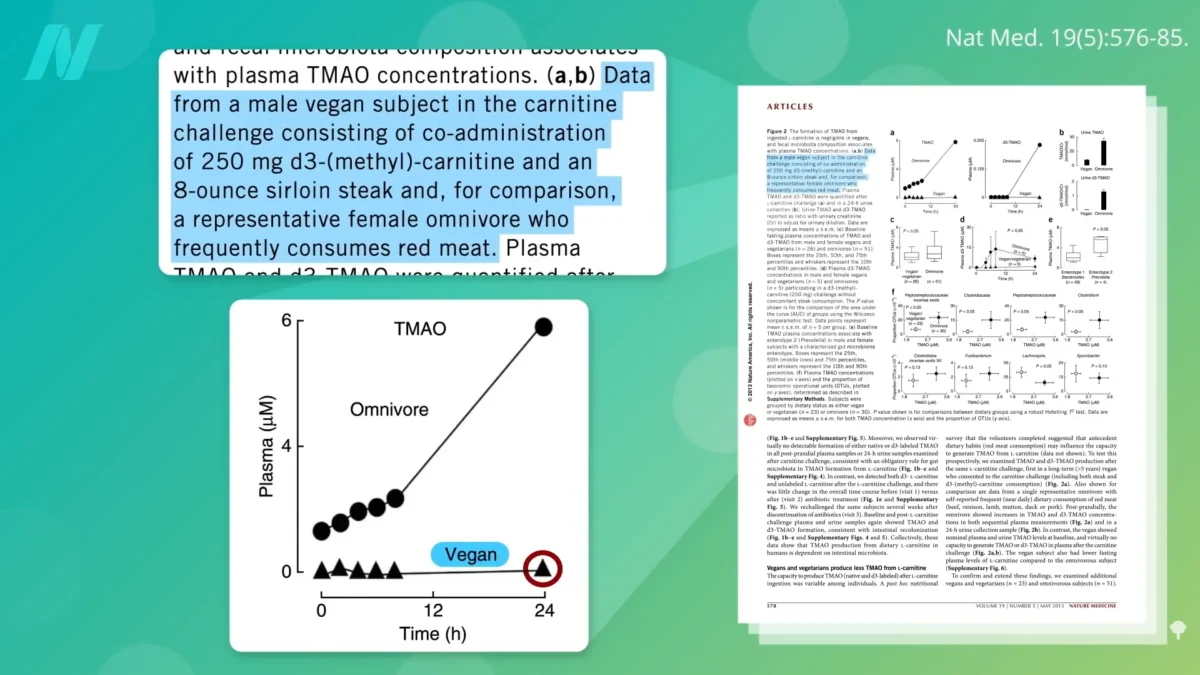
This research underscores a fundamental principle: the gut microbiome is not a static entity but a dynamic ecosystem deeply influenced by diet. When individuals adopt a plant-based diet, their gut bacteria adapt over time to efficiently process plant-derived fibers and produce beneficial metabolites. This adaptation appears to be a robust defense mechanism against the detrimental effects of TMAO. Studies have shown that even when long-term vegans are given a significant amount of carnitine-rich food, such as a 20-ounce steak, their bodies produce remarkably little TMAO compared to meat-eaters. This suggests that their gut flora has been cultivated over time to favor different metabolic pathways, making them inherently less prone to TMAO production.
The vegan fecal transplant study, in this light, highlights that there may be no quick fixes or shortcuts to achieving a TMAO-protective microbiome. The ability of "veggie feces" to mitigate TMAO production is nearly nonexistent in individuals who have consistently consumed a plant-based diet. The attempt to bypass the dietary shift through a fecal transplant, while scientifically innovative, ultimately demonstrated the primacy of diet in shaping gut health.
Broader Implications and Future Directions
The research into TMAO and its mitigation strategies has significant implications for public health. As our understanding of the microbiome’s role in chronic disease continues to expand, interventions targeting this internal ecosystem are likely to become increasingly important. However, the current evidence strongly suggests that the most effective and sustainable approach to managing TMAO levels lies in dietary modifications.
The study’s findings serve as a compelling reminder that while scientific interventions can offer insights, they cannot entirely replace foundational lifestyle choices. The journey towards a healthier gut and reduced TMAO levels appears to be intrinsically linked to adopting a diet rich in whole plant foods. This approach not only starves the bacteria that produce TMAO but also nourishes the beneficial microbes that contribute to overall well-being.
For individuals interested in exploring their own gut health and potential donor opportunities, resources are available. The concept of fecal transplants, while not a direct solution for TMAO reduction without dietary change, remains an active area of research for other conditions. Information on how to become a fecal transplant donor is accessible, and further exploration into the microbiome topic can provide deeper insights into the complex interplay between our diet, our gut bacteria, and our health.
In conclusion, while the prospect of "swapping gut flora" for immediate TMAO reduction through fecal transplantation has shown limited success in isolation, the underlying research has reinforced a crucial message: the power to influence our microbiome and, consequently, our health, lies significantly within our dietary choices. The long-term cultivation of a plant-aligned gut microbiome through sustained dietary habits remains the most potent strategy for mitigating the pervasive risks associated with TMAO.






