
The clinical landscape of men’s health is currently facing a paradoxical crisis: while diagnostic technology has never been more sophisticated, the incidence of advanced and metastatic prostate cancer is surging across North America. Despite the widespread availability of the Prostate-Specific Antigen (PSA) test, recent data indicates that the progress made in the late 20th century toward reducing prostate cancer mortality has stalled, and in some demographics, reversed. This trend is largely attributed to a fundamental shift in screening guidelines enacted over a decade ago, which prioritized the avoidance of overdiagnosis at the expense of early detection. As medical professionals and researchers reexamine the evidence, a compelling case is emerging for a modernized, multi-layered approach to screening that utilizes longitudinal data, advanced imaging, and refined biopsy techniques to eliminate preventable deaths.
The Statistical Reality of a Growing Public Health Challenge
Prostate cancer remains one of the most prevalent and lethal malignancies affecting men today. In the United States, approximately one in eight men will receive a diagnosis during their lifetime. Current projections suggest that in this year alone, roughly 36,000 American men will succumb to the disease. The tragedy of these statistics lies in the biological nature of the cancer itself; when identified in its localized stages, the prognosis is exceptionally positive, with survival rates near 100 percent. However, once the disease progresses to Stage IV—the metastatic phase where cancer spreads to distant organs or the bone—the clinical picture darkens significantly. The five-year survival rate for Stage IV prostate cancer drops to approximately 38 percent, with a median time from diagnosis to death of only 2.5 years.

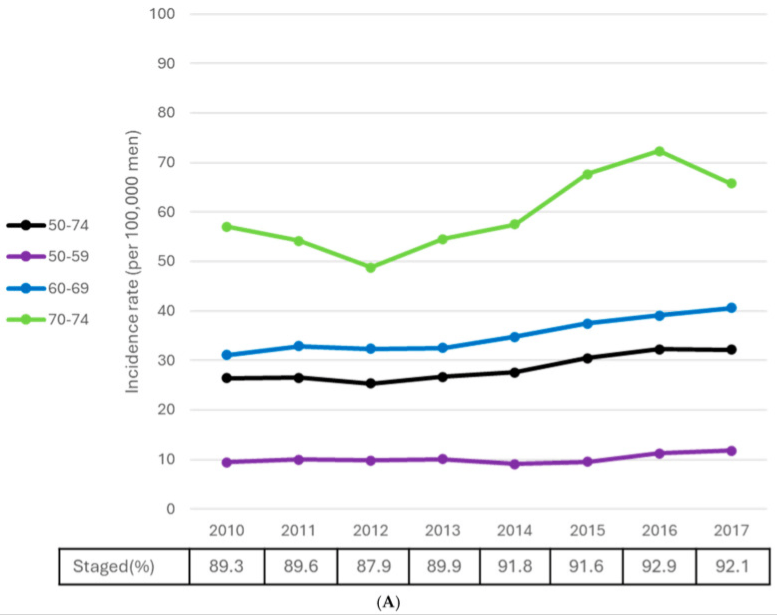
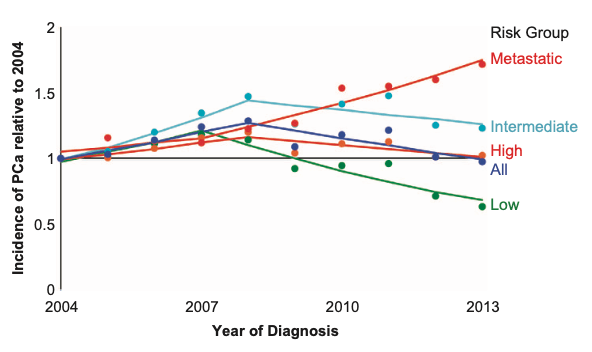
Alarmingly, the rate at which advanced prostate cancers are being diagnosed is higher today than it was 15 years ago. Data from both the United States and Canada reflect a troubling trend: we are no longer finding the disease in its curable infancy. Instead, a growing number of patients are presenting with "de novo" metastatic disease, meaning the cancer has already breached the confines of the prostate by the time it is first detected. This shift suggests a systemic failure in the early detection pipeline, leaving a generation of men vulnerable to a disease that was previously being managed with greater success.
The 2012 Guideline Shift: A Chronology of Caution
To understand the current rise in metastatic cases, it is necessary to trace the history of screening recommendations. In the 1990s and early 2000s, the PSA test was widely adopted as a frontline screening tool. The logic was straightforward: PSA is an enzyme produced by the prostate, and elevated levels in the blood often correlate with an enlarged or malignant prostate. However, the initial implementation of the test was relatively crude. Physicians often relied on single "snapshot" values, and any reading above a generic threshold (typically 4.0 ng/mL) frequently led directly to a transrectal biopsy.
By 2012, the U.S. Preventive Services Task Force (USPSTF) issued a controversial "Grade D" recommendation against routine PSA screening for all men. This decision was driven by concerns over "overdiagnosis" and "overtreatment." Because many prostate cancers are indolent—meaning they grow so slowly they are unlikely to cause symptoms or death during a patient’s natural lifespan—the USPSTF argued that the harms of screening outweighed the benefits. These harms included the physical complications of biopsies, such as sepsis and urinary incontinence, as well as the psychological and physical toll of unnecessary surgeries or radiation for low-risk tumors.
The USPSTF’s calculus relied heavily on the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial, which at the time appeared to show that PSA screening did not significantly reduce mortality. Following these guidelines, many primary care physicians stopped ordering PSA tests, leading to the decade-long decline in early detection that researchers are now linking to the current spike in Stage IV diagnoses.
Reevaluating the Evidence: The Flaw in the PLCO Trial
In the years following the 2012 guideline change, researchers began to uncover significant flaws in the data used to justify the move away from screening. In 2016, a critical analysis published in the New England Journal of Medicine revealed that the "control group" in the PLCO trial—the group that was supposedly not being screened—had actually undergone extensive PSA testing outside of the trial. Specifically, roughly 80 to 90 percent of the men in the control arm had received at least one PSA test during the study period.
This "data contamination" meant that the PLCO trial was not comparing screening to no screening; it was comparing organized screening to opportunistic screening. When researchers re-analyzed the data in 2017 using more rigorous modeling to account for this contamination, the results changed dramatically. The revised analysis suggested that PSA screening actually prevents between 25 and 33 percent of prostate cancer deaths. Despite this revelation, official guidelines from organizations like the USPSTF and the Canadian Task Force on Preventive Health Care have been slow to reflect this corrected understanding, leaving a gap between current clinical evidence and official policy.
The Modern Paradigm: PSA Velocity and Density
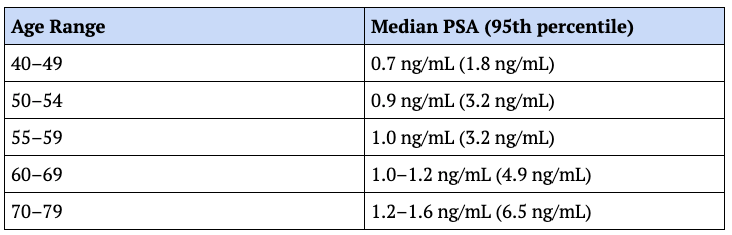
While the old method of PSA screening was undoubtedly flawed, the modern approach offers a much higher degree of precision. Experts now argue that PSA is most valuable when viewed as a longitudinal trend rather than a single value. This concept, known as "PSA Velocity," tracks how quickly a man’s PSA levels rise over time. A sustained rise of more than 0.35 ng/mL per year over an 18-month period is considered a significant "red flag," even if the absolute value remains within what is traditionally considered the "normal" range.
Furthermore, the integration of imaging has revolutionized the diagnostic process. By using Magnetic Resonance Imaging (MRI), clinicians can calculate "PSA Density"—the PSA level relative to the volume of the prostate gland. A large prostate naturally produces more PSA; therefore, a high PSA in a large gland may be benign, whereas a moderately elevated PSA in a very small gland is highly suspicious. This calculation allows doctors to filter out "false alarms" caused by benign prostatic hyperplasia (BPH) and focus on cases where the PSA production is disproportionate to the tissue volume.
Technological Advancements in Imaging and Biopsy
The diagnostic toolkit has expanded further with the introduction of biparametric MRI. Traditionally, multiparametric MRIs required the use of contrast agents (gadolinium), which added cost, time, and potential side effects to the procedure. However, the 2025 PRIME trial demonstrated that biparametric MRI—which does not require contrast—is just as effective at detecting clinically significant prostate cancer. This makes high-resolution screening more accessible and less invasive for the patient.
When a biopsy is deemed necessary, the method of collection has also evolved. The traditional transrectal approach, which involves passing a needle through the rectal wall, carries a significant risk of infection, with sepsis rates reported between 5 and 7 percent. The modern alternative is the transperineal biopsy, which accesses the prostate through the skin between the scrotum and the anus. Research, including the PREVENT trial, has shown that the transperineal approach results in near-zero infection rates and is more effective at sampling the anterior portion of the prostate, where aggressive cancers are often missed by transrectal needles.
Addressing Overtreatment Through Active Surveillance
One of the primary historical arguments against screening was that it led to unnecessary treatment. Today, the medical community has largely solved this through "Active Surveillance." Using Gleason scoring—a system that grades the malignancy of cancer cells from 1 to 5—pathologists can identify Grade Group 1 and Grade Group 2 tumors that are unlikely to spread.
Instead of immediate surgery, these patients are monitored closely with regular PSA tests and follow-up MRIs. This allows clinicians to "watch and wait," intervening only if the cancer shows signs of becoming more aggressive. By separating the diagnosis of cancer from the treatment of cancer, the modern medical community can reap the benefits of early detection without the historical baggage of overtreatment for low-risk cases.
The Finasteride Factor: A Hidden Risk in Screening
A critical but often overlooked aspect of modern prostate cancer screening is the impact of 5α-reductase inhibitors, such as finasteride and dutasteride. These medications are widely prescribed for hair loss and BPH. Mechanistically, these drugs shrink the prostate and, as a result, artificially lower PSA levels.
Clinical data suggests that after one year of finasteride use, a man’s PSA level is typically cut in half. If a clinician is unaware that a patient is taking these drugs, they may interpret a "low" PSA reading as a sign of health, when in reality, the corrected value would be twice as high. Experts recommend a correction factor: for men on finasteride for more than two years, the measured PSA should be multiplied by 2.3 or 2.5 to reflect the true biological signal. Failure to account for this can lead to a "masking" effect, where aggressive cancers are allowed to grow undetected because the chemical suppression of PSA provides a false sense of security.
Conclusion: Toward the Eradication of Prostate Cancer Mortality
The rising rates of Stage IV prostate cancer serve as a stark reminder that the current hands-off approach to screening is failing a significant portion of the population. The evidence suggests that the tools to prevent these deaths already exist; the barrier is no longer technology, but rather outdated guidelines and a lack of awareness regarding modern diagnostic protocols.
By shifting toward a personalized screening model—one that emphasizes PSA velocity, incorporates MRI-derived density, utilizes safer transperineal biopsies, and employs active surveillance for low-risk cases—the medical community has the potential to transform prostate cancer from a lethal threat into a manageable condition. As the discrepancy between rising mortality and available technology becomes clearer, the call for a national reevaluation of screening guidelines grows louder. The goal is no longer just to find cancer, but to find the right cancer at the right time, ultimately ensuring that no man dies from a disease that could have been cured if caught in its infancy.








