The landscape of weight management, particularly for individuals battling type 2 diabetes, has long been dominated by discussions of bariatric surgery. Hailed for its remarkable capacity to induce diabetes remission, procedures like gastric bypass have become a cornerstone of treatment for many. However, emerging research and a deeper understanding of the physiological mechanisms at play are challenging conventional wisdom, suggesting that the profound benefits observed post-surgery may be more closely linked to intense caloric restriction than to the anatomical alterations themselves. This shift in perspective carries significant implications for patient care, treatment accessibility, and the very definition of metabolic health.
The Promise and Evolution of Bariatric Surgery
For decades, bariatric surgery has been presented as a transformative intervention for severe obesity and its associated comorbidities, most notably type 2 diabetes. The surgical community, recognizing the profound metabolic impact of these procedures, has actively sought to reframe their perception. Moving away from terms like "internal jaw wiring" or "cutting into healthy organs," the field has embraced the designation "metabolic surgery." This nomenclature underscores the belief that surgically rearranging the gastrointestinal tract triggers significant hormonal changes that contribute to improved metabolic health.
The evidence supporting this claim has historically been compelling. Studies have consistently demonstrated high rates of type 2 diabetes remission following bariatric surgery. For instance, approximately 50% of obese individuals with diabetes and a striking 75% of those classified as "super-obese" can achieve remission—defined as normal blood sugar levels without the need for diabetes medication—after undergoing these procedures. The speed of this improvement is also noteworthy, with some patients experiencing normalized blood sugar levels within days of surgery. Long-term follow-up studies, such as one indicating that 30% of patients remained diabetes-free 15 years post-surgery compared to just 7% in a non-surgical control group, have further bolstered the perception of surgery as a definitive solution.
Questioning the Surgical Mechanism: The Liver’s Crucial Role
Despite these impressive statistics, a critical question has begun to surface: To what extent are these positive outcomes attributable to the surgery itself, and to what extent are they a byproduct of the drastic dietary changes mandated before and after the procedure? The surgical process, particularly bariatric surgery, is not without its complexities and risks. A significant challenge often encountered is the need to lift the liver, which is frequently enlarged and fatty in obese individuals. This can increase the risk of liver injury and bleeding, and in some cases, necessitates a transition from minimally invasive laparoscopic surgery to more invasive open procedures, resulting in larger scars, increased infection risk, and extended recovery times.
The pre-operative dietary protocols for bariatric surgery are designed, in part, to reduce liver size and improve surgical safety. Patients are typically placed on extremely low-calorie liquid diets for several weeks prior to surgery. This intense caloric restriction leads to rapid weight loss, and a reduction of just 5% in body weight can decrease liver fat by as much as 10%. This observation raises a pivotal question: Could the observed improvements in blood sugar levels and diabetes remission be primarily a consequence of this severe caloric deficit, rather than a unique metabolic benefit conferred by the anatomical rearrangement?

A Groundbreaking Study: Isolating Caloric Restriction from Surgery
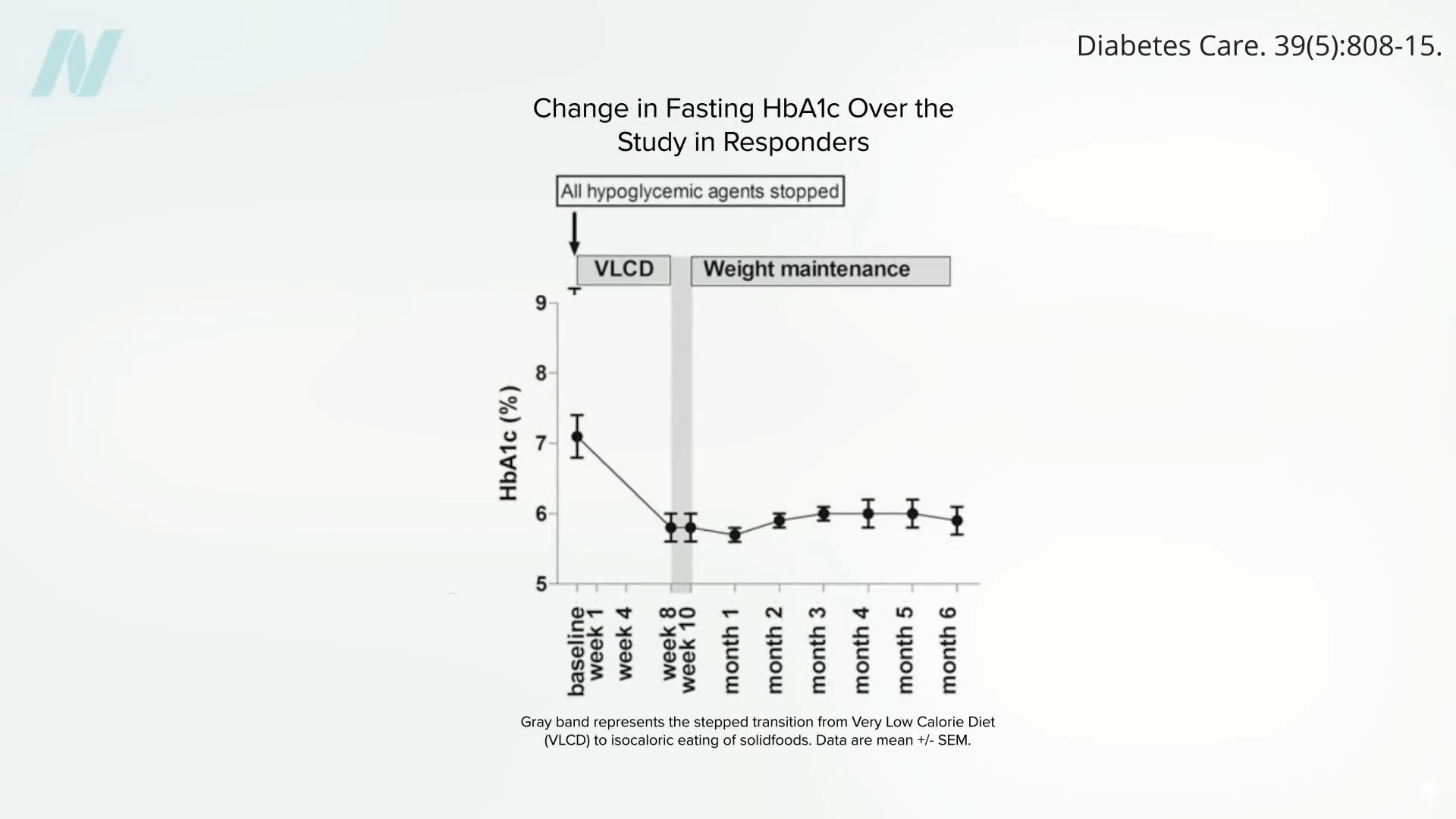
To address this critical question, researchers at a bariatric surgery clinic at the University of Texas designed a novel study. They enrolled patients with type 2 diabetes who were scheduled for gastric bypass surgery. These participants volunteered to be admitted to the hospital for a 10-day period to strictly adhere to the same extremely low-calorie diet (less than 500 calories per day) that would typically be prescribed before and after surgery. Crucially, these patients did not undergo the surgical procedure during this initial phase.
After a period of several months, during which the patients likely regained some of the lost weight, they then proceeded with their scheduled gastric bypass surgery. In a carefully controlled follow-up, they repeated the same extremely low-calorie diet, meticulously matched day-by-day with their initial dietary phase. This ingenious study design allowed researchers to directly compare the effects of caloric restriction with and without the surgical intervention, using the same patients and the same dietary regimen. The underlying hypothesis was that if the anatomical rearrangement of the gastrointestinal tract provided a distinct metabolic advantage, patients would demonstrate superior outcomes after the surgery. However, the findings revealed a surprising and significant divergence.
The Data Unveiled: Caloric Restriction’s Dominant Role
The results of this comparative study were profound. The researchers found that caloric restriction alone, independent of surgery, led to comparable improvements in blood sugar levels, pancreatic function, and insulin sensitivity. In fact, on several key measures of diabetic control, patients actually performed better without undergoing the surgery. This suggested that the surgery, in some metabolic aspects, might even place patients at a disadvantage.
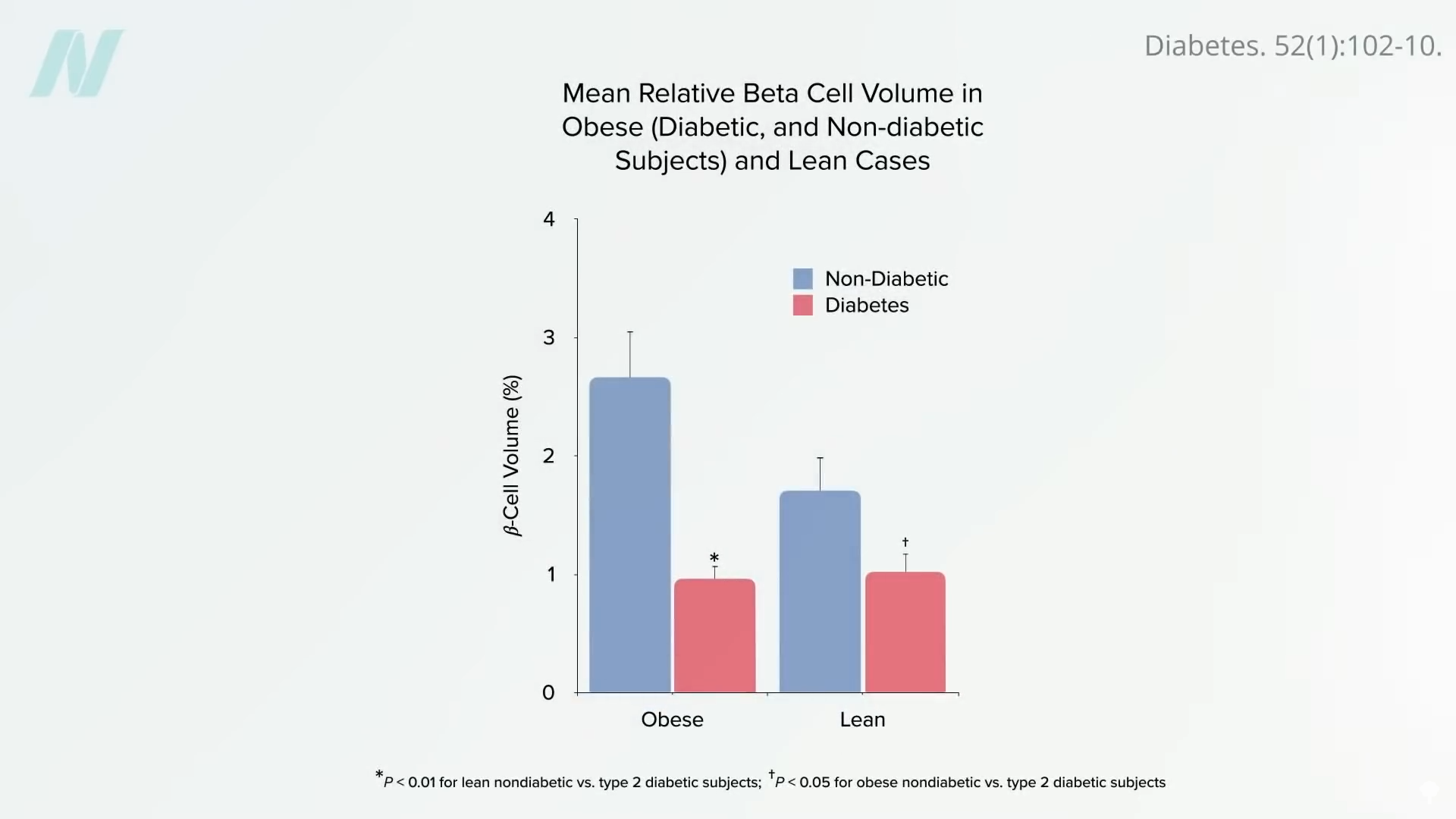
This finding aligns with a growing understanding of the pathophysiology of type 2 diabetes. The prevailing theory posits that the disease is initiated and sustained by the accumulation of excess fat within the liver. This hepatic fat accumulation leads to insulin resistance, a core problem in type 2 diabetes. When the liver’s capacity to store fat is exceeded, it begins to release fat into the bloodstream in the form of very-low-density lipoprotein (VLDL). This excess fat can then accumulate in the pancreas, damaging the insulin-producing beta cells. By the time type 2 diabetes is diagnosed, it is estimated that up to half of these crucial insulin-producing cells may have already been destroyed.
The study’s findings indicate that a sufficiently large caloric deficit can effectively reverse this process. By mobilizing fat out of the liver, a profound drop in liver fat can be achieved, leading to a resurrection of liver insulin sensitivity within a remarkably short period, sometimes as little as seven days. Sustained caloric restriction can then further reduce liver fat, contributing to the normalization of pancreatic fat levels and function within approximately eight weeks. Once an individual drops below their "personal fat threshold"—the point at which excess fat begins to negatively impact organ function—they can often resume normal caloric intake and maintain their diabetes remission.
Implications for Treatment Strategies and Patient Access
The implications of these findings are far-reaching. They suggest that the remarkable diabetes remission rates historically attributed solely to bariatric surgery may be significantly influenced, if not primarily driven, by the intensive caloric restriction that accompanies these procedures. This has several critical implications for how we approach the treatment of type 2 diabetes and obesity.
Firstly, it opens the door for less invasive and potentially more accessible treatment modalities. If the primary driver of remission is caloric deficit, then structured, medically supervised very-low-calorie diets (VCLDs) could offer a comparable, or even superior, alternative for many patients. This approach bypasses the surgical risks, the substantial costs associated with surgery, and the extended recovery periods.
Secondly, it underscores the importance of patient education and empowerment. Understanding that significant metabolic improvements are achievable through dietary interventions can motivate individuals to pursue lifestyle changes with greater conviction. This is particularly relevant for individuals who may be hesitant or unable to undergo surgery due to health concerns, financial limitations, or personal preferences.
Thirdly, it prompts a re-evaluation of post-surgical care. While bariatric surgery undeniably facilitates weight loss and can improve metabolic markers, the data suggests that continued adherence to healthy dietary principles is paramount, regardless of whether surgery was performed. The study’s finding that weight loss achieved through surgery did not necessarily improve markers of systemic inflammation, and in some cases worsened them, compared to diet-alone weight loss, highlights this nuance.
Beyond Diabetes Remission: Broader Health Benefits
The benefits of weight loss through diet alone extend beyond blood sugar control. Individuals who achieve significant weight loss through dietary interventions have also demonstrated marked improvements in systemic inflammation markers, such as tumor necrosis factor. This is a crucial consideration, as chronic inflammation is a significant contributor to a wide range of diseases, including cardiovascular disease and certain cancers.
Furthermore, the study raises questions about the long-term impact of bariatric surgery on diabetic complications, such as vision loss and kidney failure. While bariatric surgery can improve kidney function, evidence suggests it may not consistently prevent the occurrence or progression of diabetic vision loss. This could be attributed to the fact that surgery primarily addresses the quantity of food intake rather than the quality of the diet. A historical parallel can be drawn to a large study published in The New England Journal of Medicine, which randomized thousands of individuals with diabetes to an intensive lifestyle program. While participants lost weight, the study was stopped prematurely due to a lack of significant reduction in mortality or heart attacks. Researchers hypothesized that this might be because participants were still consuming a "heart-clogging" diet, albeit in smaller portions.
The Reversibility of Type 2 Diabetes: A Matter of Timing and Effort
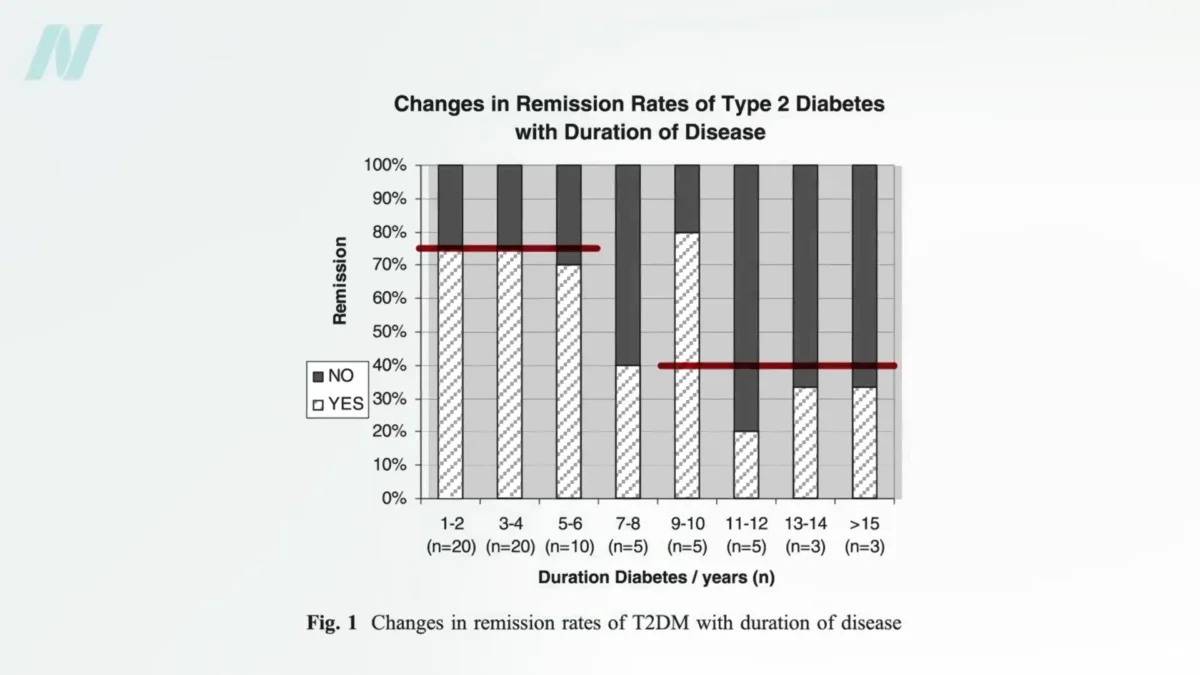
The overarching message from this evolving body of research is clear: Type 2 diabetes is a potentially reversible condition, especially when caught early. The degree of reversibility appears to be strongly correlated with the duration of the disease and the amount of weight lost. Studies indicate that individuals who have had type 2 diabetes for less than four years and lose more than 30 pounds (approximately 13.6 kilograms) through diet alone have a nearly 90% chance of achieving non-diabetic blood sugar levels. However, for those who have lived with the disease for eight or more years, this remission rate drops to around 50%.
While bariatric surgery can facilitate greater weight loss, its remission rates for diabetes do not always surpass those achieved through diet alone, particularly in the longer term. For individuals with diabetes for up to six years, remission rates with bariatric surgery hover around 75%, and for those with longer-standing diabetes, it falls to about 40%. This data further emphasizes that the effectiveness of any weight loss intervention, surgical or otherwise, in reversing type 2 diabetes is intricately linked to the underlying metabolic state and the duration of the disease.
Doctor’s Note and Future Directions
This exploration into the efficacy of bariatric surgery versus dietary interventions is part of a broader discussion on weight management strategies. The complexities of bariatric surgery, including its mortality rates and potential complications, have been detailed in previous analyses. The focus on sustainable weight loss, as explored in comprehensive resources like the book "How Not to Diet," highlights the critical role of long-term dietary habits in maintaining health and preventing chronic diseases.
The scientific community’s ongoing investigation into the mechanisms of diabetes remission is crucial. By disentangling the effects of surgical intervention from the powerful impact of caloric restriction, future treatment guidelines can be refined to offer the most effective, safe, and accessible options for individuals living with obesity and type 2 diabetes. The future of metabolic health management likely lies in a personalized approach that leverages the profound benefits of dietary interventions, potentially augmented by surgical options when appropriate, but always prioritizing the fundamental principles of nutritional science. The emphasis is shifting from simply altering anatomy to fundamentally changing metabolic processes through a controlled and sustainable reduction in caloric intake, offering a beacon of hope for improved health outcomes for millions.






