The ancient wisdom of Hippocrates, who proclaimed nearly 2,500 years ago that "All disease begins in the gut," continues to resonate with profound implications for modern health. Emerging research is increasingly highlighting the intricate relationship between our gut microbiome – the trillions of microorganisms residing within us – and a wide spectrum of diseases. A key focus of this research is Trimethylamine N-oxide (TMAO), a compound produced by gut bacteria from dietary sources, and its potential role in various health conditions. While a plant-based diet appears to confer protection against TMAO’s detrimental effects, a compelling question arises: could the direct transplantation of gut flora offer a similar protective shield?
The Double-Edged Sword of Gut Bacteria: From Beneficial Compounds to Detrimental Toxins
Our gut bacteria are not merely passive inhabitants; they are active participants in our physiology. When we nourish them with a diet rich in whole plant foods, they reciprocate by producing beneficial compounds, such as butyrate. Butyrate, a short-chain fatty acid, is a primary energy source for the cells lining our colon and plays a crucial role in maintaining gut barrier integrity and reducing inflammation. This symbiotic relationship underscores the power of "feeding our gut bacteria right."
Conversely, when the gut is fed with an abundance of certain dietary components, the microbial inhabitants can produce harmful substances. TMAO is a prime example. It is synthesized in the liver from trimethylamine (TMA), a precursor molecule generated by specific gut bacteria that metabolize compounds found abundantly in animal products, including red meat, eggs, and seafood. These foods are rich in choline and L-carnitine, nutrients that, when consumed by individuals with a particular gut microbial composition, can lead to elevated TMAO levels.
TMAO’s Expanding Association with Disease: Beyond Cardiovascular Health
Historically, TMAO was primarily linked to cardiovascular diseases. Studies have demonstrated its contribution to the development of atherosclerosis, the hardening and narrowing of arteries, thereby increasing the risk of heart attack and stroke. The mechanism involves TMAO promoting inflammation, enhancing cholesterol accumulation in artery walls, and potentially contributing to the formation of blood clots.
However, the scientific understanding of TMAO’s impact has broadened significantly in recent years. Its association is no longer confined to the cardiovascular system. Emerging research has identified links between elevated TMAO levels and a range of other chronic conditions:
- Psoriatic Arthritis: This autoimmune condition, characterized by joint inflammation and skin lesions, has been found to correlate with higher TMAO levels, suggesting a potential inflammatory pathway influenced by gut bacteria.
- Polycystic Ovary Syndrome (PCOS): This complex hormonal disorder in women has also shown an association with increased TMAO, hinting at a broader metabolic influence.
- Cancer: Perhaps most concerning is the growing body of evidence linking TMAO to increased risks of various cancers, which represent the second leading cause of death in the United States. While the precise mechanisms are still under investigation, potential pathways include TMAO-induced inflammation, oxidative stress (damage from free radicals), DNA damage, and disruptions in protein folding, all of which can contribute to cellular dysfunction and cancerous growth.
- Chronic Obstructive Pulmonary Disease (COPD): As the fourth leading killer in the U.S., COPD, including emphysema, has also shown associations with TMAO. While TMAO may not be a direct cause of COPD exacerbations, it is suspected to contribute to premature death in patients by exacerbating underlying cardiovascular issues, which are often prevalent in COPD sufferers.
- Stroke: The link between TMAO and stroke is considered a "no-brainer," a testament to its clear impact on vascular health. Elevated TMAO levels are associated with higher blood pressure, a significant risk factor for stroke. Furthermore, individuals with higher TMAO concentrations appear more prone to blood clot formation, particularly in those with atrial fibrillation, further increasing stroke risk. Studies indicate that those with higher TMAO levels not only experience more severe strokes but also face a fourfold increased risk of mortality following such an event.
- Alzheimer’s Disease: The sixth leading cause of death, Alzheimer’s disease, is also showing a connection to TMAO. TMAO has been detected in human cerebrospinal fluid, the fluid that bathes the brain. Research indicates higher TMAO levels in individuals with mild cognitive impairment and Alzheimer’s disease dementia. Within the brain, TMAO has been demonstrated to induce neuronal senescence (age-related cellular deterioration), increase oxidative stress, impair mitochondrial function (the powerhouses of cells), and inhibit mTOR signaling, all of which are implicated in brain aging and cognitive decline.
- Diabetes: As the seventh leading killer, diabetes exhibits a notable association with TMAO. Individuals with higher TMAO blood levels are approximately 50% more likely to develop diabetes.
- Pneumonia: This eighth leading cause of death can also be influenced by TMAO. TMAO levels have been found to predict fatal outcomes in pneumonia patients, even in the absence of overt heart disease.
- Kidney Disease: Occupying the ninth position among leading causes of death, kidney disease shows a strong relationship with TMAO. TMAO levels are closely linked to kidney function and can predict fatal outcomes. A five-year study revealed that over half of chronic kidney disease patients with average or higher TMAO levels died, while nearly 90% of those in the lowest third of TMAO levels remained alive, underscoring TMAO’s prognostic value in kidney health.
Exploring Strategies to Mitigate TMAO Levels
Given the pervasive negative health associations of TMAO, a critical question emerges: how can individuals effectively lower their blood TMAO levels? Since TMAO originates from dietary precursors, a primary strategy involves reducing the intake of foods rich in choline and L-carnitine. These nutrients are abundant in animal products like meat, eggs, and dairy.
However, the widespread presence of these compounds in common dietary staples makes strict restriction challenging for many. This practicality issue has led researchers to explore more innovative interventions, including the concept of altering the gut microbiome itself.
The Promise and Pitfalls of Fecal Microbiota Transplantation (FMT)
The idea of directly transferring gut flora from a healthy donor to a recipient is known as Fecal Microbiota Transplantation (FMT). This approach has shown significant success in treating recurrent Clostridioides difficile infections. The question then becomes: could a "vegan fecal transplant" – a transplant from a donor who adheres to a strict plant-based diet – effectively lower TMAO levels in recipients?
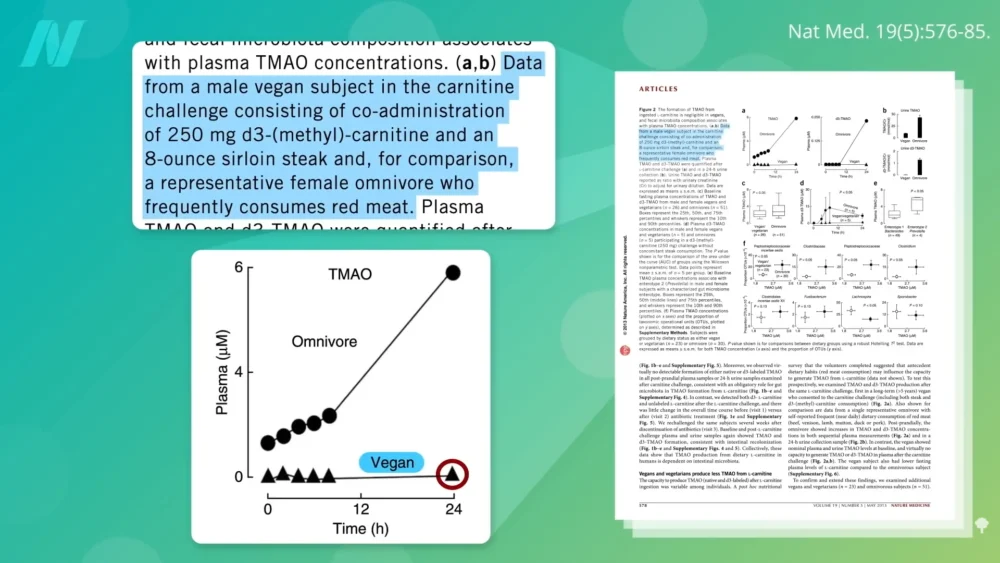
Studies have provided intriguing insights into the gut microbiome’s adaptation to diet. For instance, individuals following a plant-based diet for an extended period tend to harbor gut bacteria that are less efficient at producing TMAO, even when they consume carnitine-rich foods. In contrast, individuals with a history of meat consumption often possess a microbiome predisposed to higher TMAO production. This suggests that the composition of gut flora is significantly influenced by long-term dietary habits.

In one notable study, even when individuals on a plant-based diet were given the equivalent of a 20-ounce steak daily for two months, only about half showed an increase in TMAO production. This indicates a remarkable resilience of the plant-based gut microbiome, requiring substantial dietary shifts to alter TMAO-producing capacity. Conversely, the capacity of fecal matter from a vegan donor to produce TMAO was observed to be almost nonexistent.
This led to the exploration of direct intervention through FMT. A double-blind, randomized, controlled trial investigated the effects of vegan fecal transplants. Participants received either vegan fecal matter or their own re-transplanted fecal matter via a nasogastric tube. However, the results of this particular trial did not demonstrate a significant improvement in TMAO levels in the recipients.
Several factors may have contributed to these findings. Firstly, the vegans recruited for this study, unlike those in some previous observational studies, were found to be producing TMAO themselves. This might be attributed to differences in the duration of their vegan diet; the earlier observational study required participants to have been vegan for at least a year, allowing for more profound microbial shifts. In the FMT trial, the vegan donors may not have had a sufficiently distinct microbiome profile to induce significant changes in recipients within the study’s timeframe. Furthermore, the vegan fecal matter used in the transplant itself might have possessed some inherent capacity to produce TMAO.
The researchers suggested that the lack of improvement post-transplant could be related to limited baseline microbiome differences between donors and recipients and, crucially, the continuation of an omnivorous diet by the recipients after the transplant. This raises a critical point: the effectiveness of any microbiome intervention, including FMT, may be significantly undermined if the recipient does not subsequently adopt a supportive diet. The researchers intentionally allowed participants to continue their omnivorous diets to isolate the effect of the transplant itself, acknowledging that a plant-based diet alone is known to alter the microbiome.
The Unavoidable Conclusion: Diet Remains Paramount
The findings from these investigations suggest that while the concept of manipulating the gut microbiome through transplantation is scientifically intriguing, it may not offer a shortcut to circumventing the fundamental impact of diet on TMAO production and its associated health consequences. The intricate interplay between diet and the gut microbiome appears to be deeply ingrained, suggesting that long-term dietary patterns are the most influential factor in shaping an individual’s TMAO profile.
In essence, the research points towards a clear, albeit perhaps less glamorous, conclusion: the most effective and sustainable strategy for managing TMAO levels and mitigating its associated health risks lies in dietary modification. Embracing a diet rich in whole plant foods, which naturally promotes a beneficial gut microbiome and limits the intake of TMAO precursors, remains the cornerstone of promoting gut health and overall well-being.
Doctor’s Note:
For individuals interested in becoming a potential donor for fecal microbiota transplantation, resources are available to guide you through the process. Information on how to become a fecal transplant super donor can be found through relevant health organizations and research institutions.
Further exploration into the complex topic of TMAO and its impact on health can be found in related publications and resources. The microbiome’s role in health and disease is a rapidly evolving field, and staying informed about the latest research is crucial for understanding and addressing the challenges it presents. The microbiome topic page offers a comprehensive overview of this fascinating area of scientific inquiry.






