Recent investigations utilizing sham surgery trials have cast a stark and disquieting light on the efficacy of some widely adopted surgical interventions, suggesting that a significant number of these procedures may offer little to no genuine therapeutic benefit, effectively rendering them comparable to sham treatments. This revelation challenges long-held assumptions about medical progress and the evidence base supporting common surgical practices, particularly in the realm of weight loss and orthopedic interventions.
The Rise and Fall (and Return) of the Intragastric Balloon
The intragastric balloon, a device designed to occupy space within the stomach and induce satiety, first emerged in the 1980s amidst considerable fanfare. Its purported simplicity – implantation into the stomach and inflation with air or water to create a feeling of fullness – positioned it as a less invasive alternative to traditional bariatric surgery. However, the history of medical devices is often marked by their introduction to the market preceding robust evidence of their safety and effectiveness, a pattern that the intragastric balloon exemplifies.
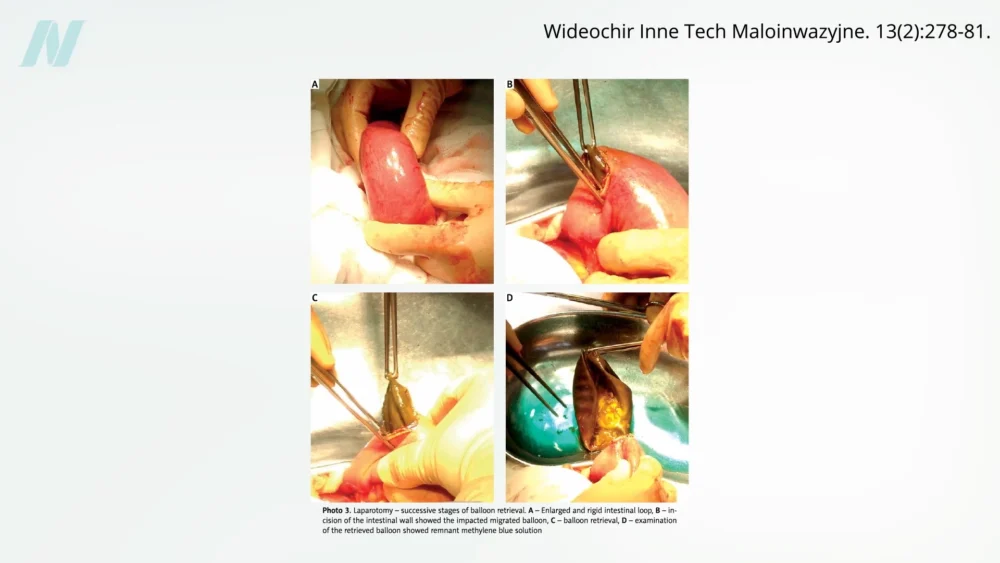
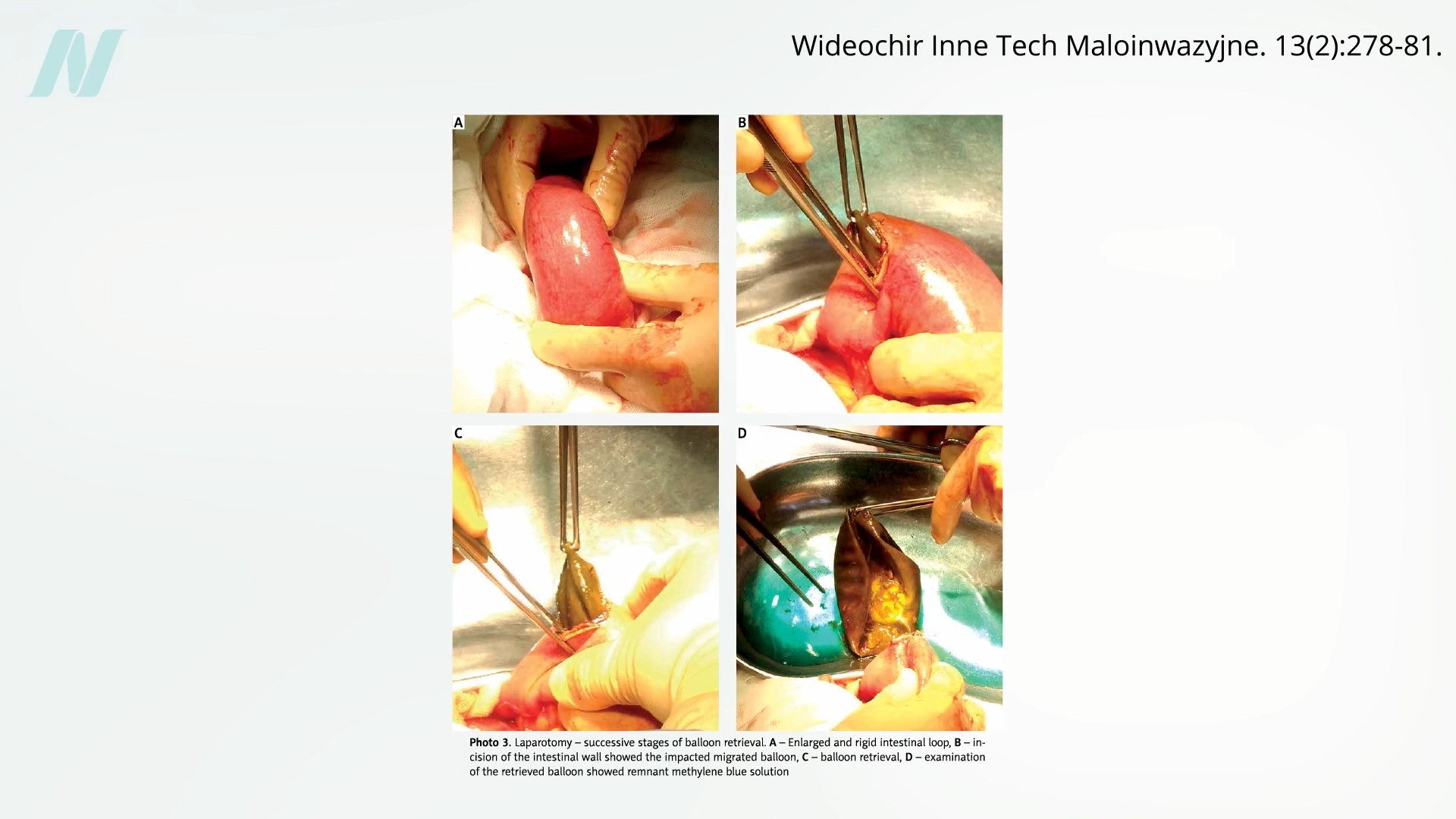
Early research soon began to question the balloon’s efficacy. A pivotal study conducted at the Mayo Clinic revealed a startling statistic: eight out of every ten intragastric balloons spontaneously deflated. This malfunction posed a significant risk, as deflated balloons could migrate into the intestines, leading to potentially life-threatening obstructions. The visual evidence, including diagrams and video footage, starkly illustrates the danger of these devices moving from their intended gastric location to the delicate intestinal tract.

Beyond the mechanical risks, the balloons also demonstrated a propensity for causing harm even when intact. Prior to deflation, these devices were found to cause gastric erosions in approximately half of the patients who received them, leading to damage of the stomach lining. More critically, when compared to non-surgical interventions such as dietary changes and behavioral modification strategies, the intragastric balloons failed to demonstrate superior effectiveness in inducing significant weight loss. This confluence of limited efficacy and potential harm ultimately led to their withdrawal from the market.
A Second Coming and Lingering Concerns
After a hiatus of 33 years, intragastric balloons were re-introduced to the U.S. market in 2015, with the Food and Drug Administration (FDA) approving a new generation of devices. This resurgence saw the placement of over 5,000 devices within a short period. The re-emergence of these devices occurred in an era shaped by legislative changes like the Sunshine Act, which mandates disclosure of financial relationships between healthcare providers and the pharmaceutical and medical device industries. This legislation aimed to shed light on potential conflicts of interest, revealing that top physicians can receive substantial payments, sometimes millions of dollars annually, from device manufacturers. Alarmingly, many of these physicians have been found to fail to disclose these financial ties when publishing research on the very devices they are financially linked to.
While proponents highlight the reversibility of intragastric balloons as a key advantage over permanent bariatric surgery, this characteristic does not render them benign. The FDA has issued a series of advisories detailing the serious risks associated with these devices, including the possibility of death. The mechanism of injury can be indirect yet devastating. Severe nausea and vomiting, common side effects affecting a majority of patients, can lead to such intense abdominal pressure that the stomach can rupture. Furthermore, persistent vomiting has been implicated in cases of life-threatening nutrient deficiencies following balloon implantation.
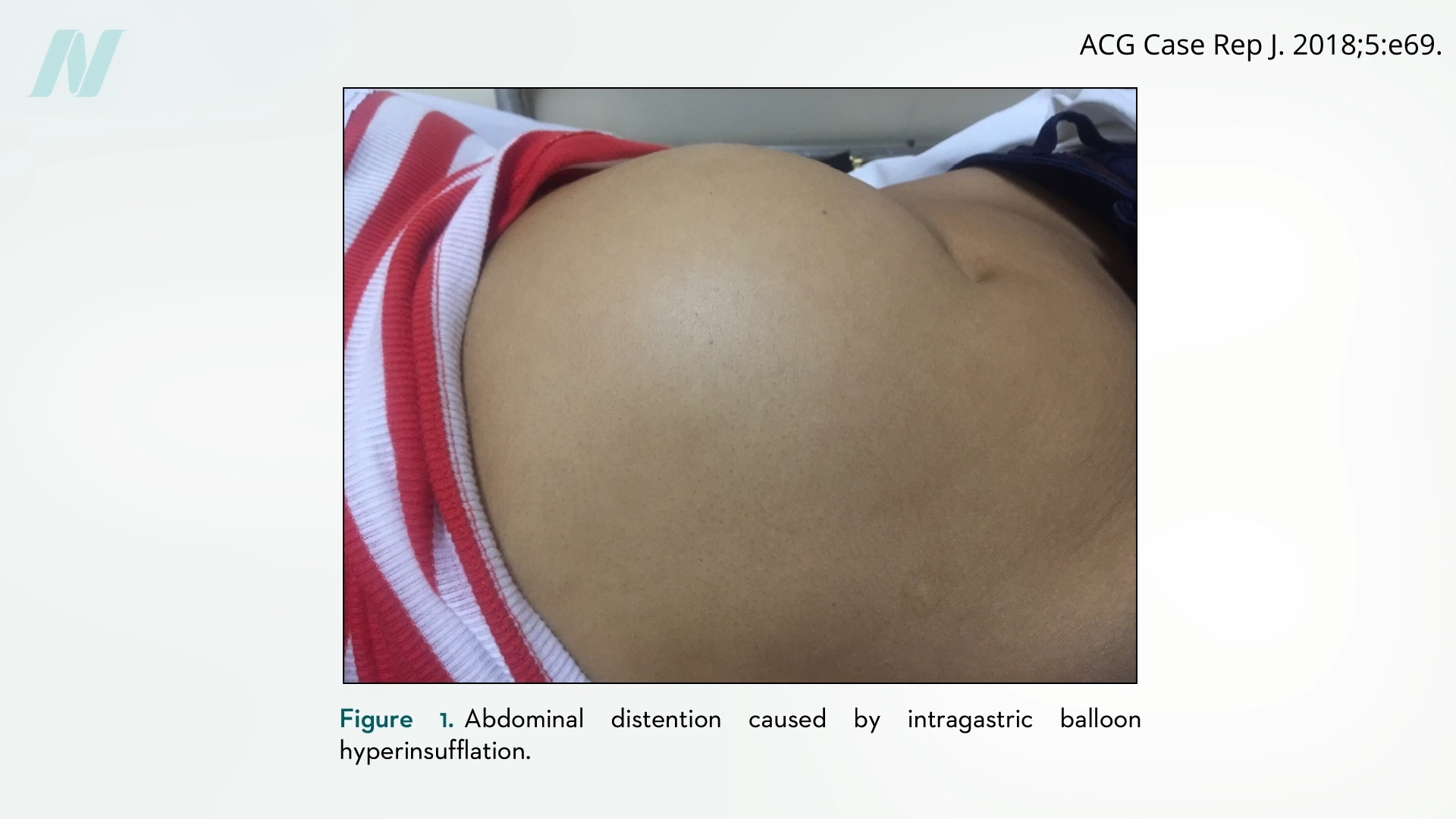
Complications are not solely attributable to deflation or rupture. Some balloons have been documented to overinflate unexpectedly, leading to severe pain, vomiting, and abdominal distention. This phenomenon, mirroring issues previously observed with spontaneously autoinflating breast implants, where implant volume can increase dramatically and unpredictably, remains an area of concern and ongoing investigation within the medical community. The historical connection between early experimental intragastric balloons and breast implant technology further underscores the evolving and sometimes problematic trajectory of medical device development.
The Enigma of Sham Surgery and Its Implications
The concept of sham surgery, while ethically controversial, has become an indispensable tool in rigorously evaluating the effectiveness of medical procedures. It involves performing a surgical procedure that mimics the actual intervention without actually performing the therapeutic part of the operation. This allows researchers to isolate the specific benefits of the surgical technique from the placebo effect, the psychological impact of undergoing a medical procedure, and the influence of accompanying supportive care.
A landmark study published in The New England Journal of Medicine in 2002 subjected arthroscopic knee surgery, a procedure performed billions of dollars annually for osteoarthritis and knee injuries, to this rigorous testing. Participants with knee pain were randomly assigned to either undergo the actual surgery or a sham procedure, where surgeons made incisions and simulated the operation without treating the joint. The study’s findings triggered an uproar within the medical community, with professional associations questioning the ethics of such trials and the consent of patients involved.
The results were startling: while patients who received the actual surgery did show improvement, so too did the placebo group. This indicated that a significant portion of the perceived benefit from arthroscopic knee surgery might be attributable to the placebo effect rather than the surgical intervention itself. Subsequent research has indicated that arthroscopic knee surgery may have no actual effect and could even increase the risk of needing a total knee replacement, a finding that has led to a crisis of confidence in the procedure. Similarly, rotator cuff shoulder surgery is currently facing similar scrutiny.
When intragastric balloons were subjected to sham-controlled trials, the results were equally revealing. Studies demonstrated that both older and newer balloon designs often failed to provide any significant weight loss benefit. Even in cases where some weight loss occurred, it was frequently temporary. This is partly because balloons are typically removed after six months, a timeframe dictated by the increasing risk of deflation. Attempts to prolong their use or to repeatedly implant new balloons have not demonstrably improved long-term weight outcomes. Sham-controlled trials have further suggested that any effects on appetite and satiety may diminish over time as the body adapts.
Broader Implications for Medical Practice and Public Trust
The findings from sham surgery trials extend beyond specific weight-loss devices and orthopedic procedures. They raise fundamental questions about the evidence base for a range of popular surgical interventions. This has led to a critical self-examination within the medical profession, which often prides itself on its scientific underpinnings. The parallel drawn between the medical community’s critique of anti-vaccination movements and the emergence of "alternative facts" within medicine itself highlights a potential hypocrisy.
The implications of these findings are far-reaching. For patients, it underscores the critical importance of seeking out procedures backed by robust, independent evidence. It also necessitates a deeper understanding of the potential conflicts of interest that can influence medical recommendations. The financial ties between physicians and device manufacturers, while now more transparent due to legislation, can still subtly influence clinical decision-making and the dissemination of research.
For the medical industry, these trials serve as a crucial reminder of the need for rigorous, unbiased research before widespread adoption of new technologies and procedures. The focus must shift from commercial expediency to patient well-being, ensuring that interventions are not only novel but also demonstrably safe and effective.
The ongoing dialogue surrounding these issues is essential for maintaining public trust in the medical system. Transparency, ethical research practices, and a commitment to evidence-based medicine are paramount. As the medical landscape continues to evolve, the lessons learned from sham surgery trials will undoubtedly shape future research priorities, regulatory oversight, and the very definition of what constitutes effective medical care. The series will continue by examining other extreme weight-loss devices, further illuminating the complex and often surprising world of medical interventions.






