Sham surgery trials, a controversial yet critical methodology in medical research, have begun to cast a stark light on the efficacy of some of the most widely performed surgical interventions. These trials, designed to rigorously isolate the true impact of a procedure from psychological effects and the benefits of active intervention, are revealing that certain popular surgeries may offer little to no significant advantage over placebo treatments. The implications of these findings are profound, questioning established medical practices, highlighting potential conflicts of interest within the healthcare industry, and demanding a re-evaluation of patient care.
The Rise and Fall, and Resurgence, of the Intragastric Balloon
One of the most prominent examples of this phenomenon involves the intragastric balloon, a device introduced in the 1980s with considerable fanfare. The concept was straightforward: implanting a deflated balloon into the stomach and then inflating it with air or water to occupy a significant portion of the stomach’s volume. This artificial fullness was intended to promote satiety, thereby aiding in weight loss. However, the history of medical devices, including the intragastric balloon, is often marked by their premature introduction to the market, preceding robust evidence of their safety and effectiveness.
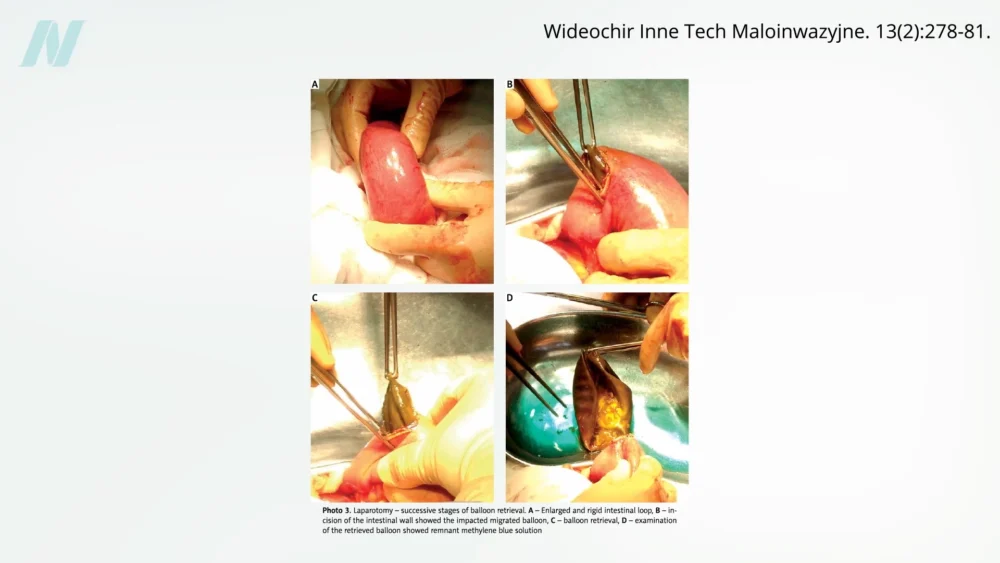
Early studies soon began to puncture the inflated expectations surrounding the "gastric bubble." A pivotal study conducted at the Mayo Clinic, published in the early 1990s, revealed a startling failure rate: 8 out of 10 intragastric balloons spontaneously deflated. This mechanical failure presented a serious and potentially life-threatening complication. Deflated balloons could migrate from the stomach into the intestines, leading to intestinal obstruction, a medical emergency requiring immediate surgical intervention. The visual evidence from case studies and video documentation underscores the potential for such blockages, a risk highlighted in patient education materials.
Beyond the mechanical failures, the intragastric balloon posed significant physiological risks even before deflation. Data indicated that approximately half of the patients experienced gastric erosions, a damaging effect on the stomach lining. Perhaps more damning was the finding that, when compared to less invasive interventions like diet and behavioral modification strategies, the intragastric balloon offered no superior weight-loss results. This lack of demonstrable efficacy, coupled with the inherent risks, ultimately led to the withdrawal of intragastric balloons from the market.

The Return of the Balloon and the Shadow of Industry Influence
After a hiatus of over three decades, intragastric balloons re-emerged in the United States. In 2015, the U.S. Food and Drug Administration (FDA) began approving a new generation of these devices. This reintroduction coincided with the passage of the Sunshine Act, a landmark piece of legislation designed to enhance transparency in financial relationships within the healthcare sector. The Sunshine Act mandates the disclosure of payments made by pharmaceutical companies and medical device manufacturers to physicians, aiming to shed light on potential conflicts of interest.
While the public is increasingly aware of the financial ties between doctors and "Big Pharma," the influence of medical device companies on surgeons often remains less visible. The financial incentives can be substantial. Reports have shown that top physician recipients of industry payments can receive millions of dollars annually from device manufacturers. Alarmingly, even when publishing research related to these devices, a significant minority of physicians have failed to disclose these potential conflicts of interest, raising questions about the objectivity of their findings.
Although intragastric balloons offer a theoretical advantage over more permanent bariatric surgeries in that they are reversible, their safety profile remains a significant concern. The FDA has issued multiple advisories detailing the serious risks associated with these devices, including the potential for death. The mechanism of injury can be counterintuitive: a smooth, rounded object causing severe and persistent vomiting. This forceful expulsion can, in rare but documented cases, lead to a rupture of the stomach. Nausea and vomiting are indeed very common side effects, affecting the majority of patients. Furthermore, persistent vomiting can contribute to life-threatening nutrient deficiencies, a consequence that can arise even after the balloon is removed.
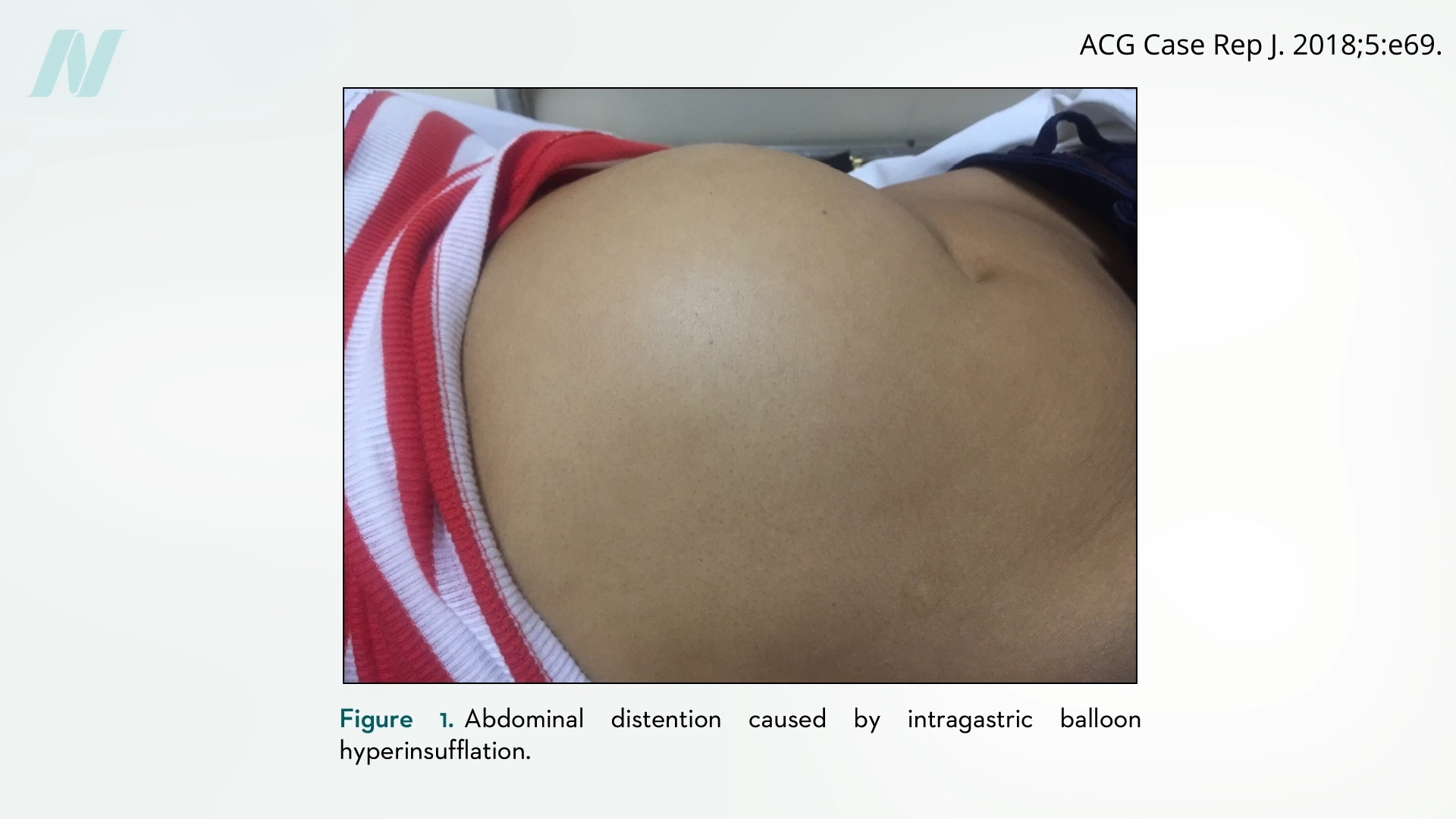
Complications associated with intragastric balloons are not limited to deflation. Some cases of bowel obstruction are directly linked to device deflation. However, an equally concerning issue is the phenomenon of spontaneous overinflation. In these instances, the balloons can unexpectedly expand beyond their intended capacity, leading to severe abdominal pain, intense vomiting, and significant abdominal distention. This unpredictable overinflation can necessitate urgent medical intervention.
The Broader Implications of Sham Surgery Trials
The unsettling patterns observed with intragastric balloons echo findings from sham surgery trials in other surgical specialties. The concept of sham surgery involves performing a surgical procedure that mimics the real operation but without the therapeutic intervention. In the context of weight-loss devices, this might involve placing a sham balloon or performing a procedure that appears to be the actual surgery but where no active treatment is delivered. The purpose is to isolate the specific effect of the intervention from the placebo effect, the psychological benefits of undergoing any medical procedure, and the impact of accompanying care like dietary counseling.
This methodology gained significant traction and controversy in 2002 with a groundbreaking study published in The New England Journal of Medicine concerning arthroscopic knee surgery. This procedure, one of the most frequently performed orthopedic surgeries globally, involves inserting a small camera and instruments into the knee joint to diagnose and treat injuries or cartilage damage. Billions of dollars are expended annually on these operations for osteoarthritis and knee injuries.
The study, however, dared to challenge this established practice. Patients suffering from knee pain were randomly assigned to one of three groups: those who received the actual arthroscopic surgery, those who underwent a sham surgery (involving an incision and simulated surgical actions, including the instillation of saline, but without any actual treatment of the joint), and a control group receiving only medication. The results of this trial ignited a firestorm within the medical community. Professional medical associations questioned the ethics of subjecting patients to sham surgery, deeming it potentially deceptive. The sanity of patients who consented to such a procedure was also debated.
The findings were stark: patients who received the actual surgery experienced improvement, but crucially, so did those who underwent the sham procedure. The outcomes for both groups were virtually indistinguishable, suggesting that the perceived benefits of arthroscopic knee surgery were largely attributable to the placebo effect rather than the surgical intervention itself. Subsequent research has indicated that in some cases, the actual surgery may even lead to worse long-term outcomes, such as an increased risk of progression to the need for a total knee replacement. This has led to a crisis of confidence regarding other common orthopedic procedures, including rotator cuff shoulder surgery.
Re-evaluating the Evidence: Weight Loss and Beyond
When intragastric balloons were subjected to sham-controlled trials, the results further corroborated the concerns. These studies consistently demonstrated that both older and newer iterations of the devices often failed to provide any meaningful or lasting weight-loss benefit. Even when some degree of weight loss was observed, it was frequently temporary. This is partly because balloons are typically intended for short-term use, generally limited to six months, due to the escalating risk of deflation. Attempts to extend usage or repeatedly implant new balloons have not yielded improved long-term weight outcomes in sham-controlled studies. These trials have suggested that any initial effects on appetite and satiety may diminish over time as the body adapts.
The implications of these sham surgery trials extend far beyond weight-loss interventions. They fundamentally challenge the scientific basis of certain widely accepted surgical practices. While the medical community rightly emphasizes evidence-based medicine and actively combats misinformation, such as the anti-vaccination movement, these studies reveal a parallel challenge within medicine itself. The tendency to "choose one’s own facts" or embrace alternative explanations is not exclusive to the public; it can manifest within medical practices when established procedures are not rigorously re-evaluated against placebo controls.
The Ethical and Financial Landscape
The existence of financial incentives from medical device companies for surgeons, coupled with the lack of disclosure in published research, creates a complex ethical and financial landscape. The Sunshine Act has brought some of this into the open, but the potential for bias in research and clinical recommendations remains a significant concern. When a procedure’s effectiveness is questionable, and its promotion is potentially influenced by financial ties, patients are put at risk.
The phenomenon of spontaneous overinflation in intragastric balloons also raises broader questions about device design and manufacturing oversight. Similar to issues previously documented with breast implants, where implants could spontaneously increase in volume, the unpredictable behavior of medical devices can have severe consequences. These underreported and poorly understood phenomena highlight the need for more stringent regulatory oversight and post-market surveillance.
The Path Forward: Transparency and Patient Empowerment
The insights gleaned from sham surgery trials are not intended to discredit all surgical interventions. Many procedures have proven efficacy and are indispensable for treating a wide range of conditions. However, these trials serve as a critical reminder that the status quo in medicine should not be accepted without rigorous scrutiny. They underscore the importance of:
- Independent Research: Prioritizing funding for independent research that is not beholden to industry interests.
- Rigorous Trial Design: Employing sham controls when ethically feasible and scientifically necessary to accurately assess the true impact of surgical interventions.
- Full Disclosure: Enforcing strict disclosure requirements for any financial or professional relationships between researchers, clinicians, and the medical device industry.
- Patient Education: Empowering patients with comprehensive and unbiased information about the risks, benefits, and uncertainties of all medical procedures.
As the medical field continues to advance, a commitment to evidence-based practice, transparency, and a critical examination of established norms is paramount. Sham surgery trials, despite their ethical controversies, offer an invaluable lens through which to ensure that the surgeries patients undergo are not merely procedures performed, but interventions that demonstrably improve health outcomes. The ongoing investigation into the effectiveness of popular medical interventions, spurred by these rigorous trials, is essential for safeguarding patient well-being and maintaining public trust in the healthcare system. The lessons learned from the intragastric balloon and knee surgery trials are a call to action for a more evidence-driven and ethically transparent approach to medical innovation and practice.






