A pioneering study led by researchers at NYU College of Dentistry has unveiled a fundamentally new strategy for tackling obesity, shifting focus from appetite suppression to significantly increasing the body’s natural energy expenditure. Published in the esteemed journal Nature Communications, the findings pinpoint a specific protein, SLIT3, as a critical orchestrator in building the essential nerve and blood vessel networks within brown fat, a specialized tissue renowned for its heat-generating capabilities. This discovery introduces the potential for therapeutic interventions that could enhance the body’s internal "metabolic sink," offering a distinct alternative to current blockbuster weight-loss medications like GLP-1s, which primarily work by reducing food intake.
A New Frontier in Weight Loss: Beyond Appetite Suppression
For decades, the battle against obesity has largely centered on caloric restriction and increasing physical activity. More recently, pharmaceutical innovations, most notably GLP-1 receptor agonists such as Ozempic and Wegovy, have revolutionized weight management by modulating hunger signals and satiety, thereby decreasing caloric intake. While highly effective for many, these drugs represent one side of the metabolic equation: reducing energy in. The NYU research proposes an entirely different approach: boosting energy out by making the body more efficient at burning calories.
Dr. Farnaz Shamsi, an assistant professor of molecular pathobiology at NYU College of Dentistry and senior author of the study, emphasized this paradigm shift. "While current weight-loss blockbusters like GLP-1s focus on suppressing appetite, our research has uncovered a completely different strategy: increasing energy expenditure by ‘building out’ the body’s natural heat-generating tissue," she explained. This novel pathway suggests that obesity could be treated by enhancing the body’s internal metabolic furnace rather than solely focusing on eating less.
The Metabolic Powerhouse: Understanding Brown Adipose Tissue
To fully appreciate the significance of this discovery, it is crucial to understand the distinct roles of different types of fat in the human body.
-
White vs. Brown Fat: A Crucial Distinction: The majority of fat in our bodies is white adipose tissue (WAT). Its primary function is to store excess energy in the form of triglycerides. While essential for insulation and hormone production, excessive accumulation of white fat is directly linked to obesity and a host of metabolic disorders, including type 2 diabetes, heart disease, and non-alcoholic fatty liver disease. In contrast, brown adipose tissue (BAT), or brown fat, is a specialized type of fat found in humans and other mammals. Unlike white fat, brown fat’s main purpose is not energy storage but thermogenesis – the generation of heat. Historically thought to be present only in infants for non-shivering thermoregulation, research over the past two decades has confirmed its presence and metabolic activity in adult humans, sparking immense interest in its therapeutic potential for obesity and metabolic health.
-
The Process of Thermogenesis: When activated, typically by exposure to cold temperatures, brown fat cells rapidly take up and metabolize glucose and lipids (fatty acids) from the bloodstream. Instead of converting these fuel sources into ATP (cellular energy) for storage, brown fat possesses a unique protein called uncoupling protein 1 (UCP1) in its mitochondria. UCP1 "uncouples" oxidative phosphorylation, diverting the energy from fuel oxidation directly into heat production, effectively burning calories without performing mechanical work. As Dr. Shamsi articulated, "During thermogenesis, all of that chemical energy is dissipated as heat instead of being stored in the body as white fat. By rapidly taking up and using fuel sources from our bodies and the food that we eat, brown fat acts like a metabolic sink that draws in nutrients and prevents them from being stored." This "metabolic sink" function makes brown fat an incredibly attractive target for interventions aimed at increasing energy expenditure.
Unraveling the SLIT3 Mechanism: Building Brown Fat’s Infrastructure
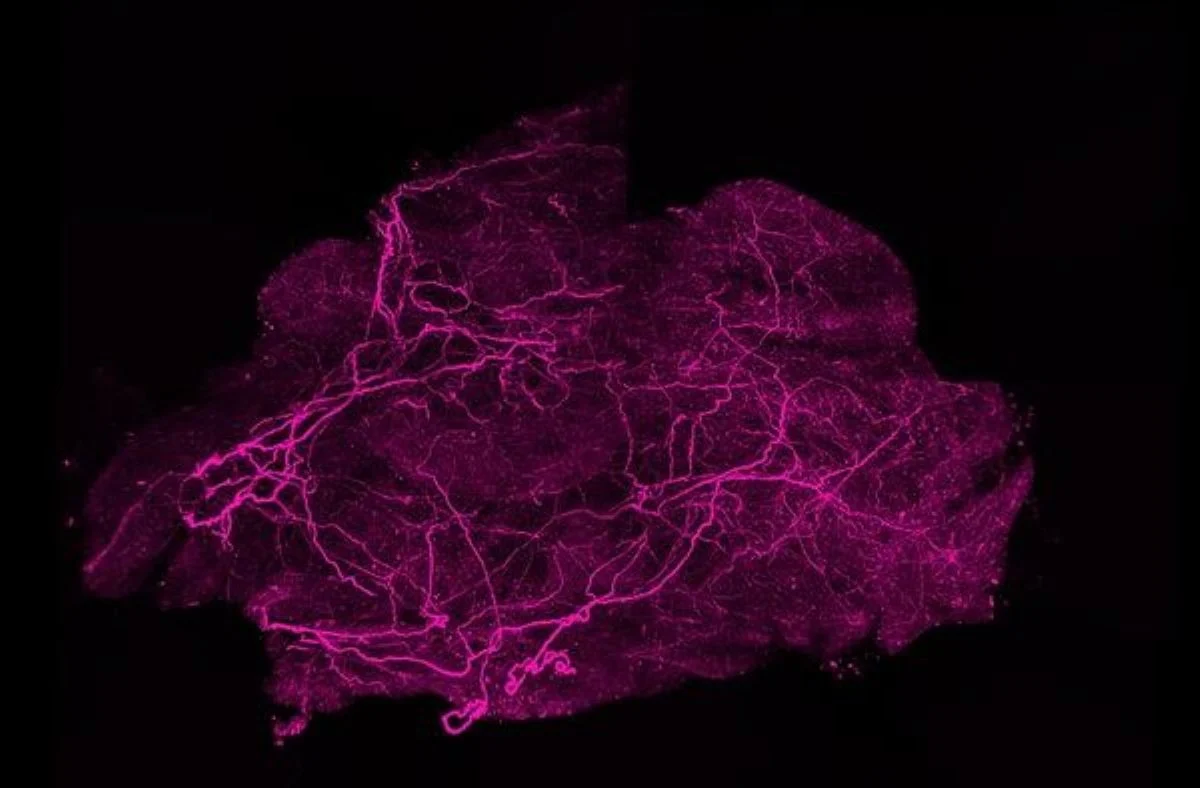
Despite the known benefits of brown fat, simply having it isn’t enough. Its ability to effectively burn calories hinges on a sophisticated internal "infrastructure" of nerves and blood vessels. These intricate networks are critical for brown fat’s optimal functioning. Nerves provide the communication link with the brain, allowing it to receive "get warm" signals, typically triggered by cold exposure. Blood vessels, on the other hand, are the supply lines, delivering the necessary oxygen and nutrients (glucose and lipids) for thermogenesis and subsequently distributing the generated heat throughout the body. Until now, much of the research on brown fat has focused on stimulating the fat cells themselves, with less attention paid to how these underlying support networks are established and maintained.
The NYU study delves into this critical infrastructure, identifying the protein SLIT3 as a key player. Dr. Shamsi’s lab had previously identified SLIT3, a protein secreted by brown fat cells, through single-cell RNA sequencing, suspecting its role in cellular communication. The current research elegantly demonstrates that when SLIT3 is produced, it undergoes a crucial cleavage process, splitting into two distinct fragments.
-
The "Split Signal" at Work: The researchers, using a combination of human and mouse cell models, identified the enzyme BMP1 as responsible for this precise cleavage of SLIT3. They then determined that these two SLIT3 fragments are not redundant but control different, yet coordinated, processes essential for brown fat activation. One fragment is responsible for promoting the growth and expansion of the blood vessel network, ensuring a robust supply of fuel and oxygen. The other fragment drives the expansion of the nerve network, which is vital for the brain to signal brown fat to become active and initiate thermogenesis. "It works as a split signal, which is an elegant evolutionary design in which two components of a single factor independently regulate distinct processes that must be tightly coordinated in space and time," Dr. Shamsi elaborated. This synchronized development of both neurovascular components is paramount for brown fat’s effective response to cold and its calorie-burning capacity. The study further identified PLXNA1 as the specific receptor that binds to one of the SLIT3 fragments, mediating its control over the nerve network in brown fat.
-
Evidence from Lab to Clinic: The robustness of these findings was established through a series of experiments. In mouse models, which naturally possess very active brown fat and exhibit high tolerance to cold, the researchers observed striking results. When SLIT3 or its receptor, PLXNA1, were genetically removed from brown fat, the mice became markedly sensitive to cold temperatures and struggled to maintain their body heat. A meticulous examination of their brown fat tissue revealed significant deficiencies: a lack of proper nerve structure and reduced density of blood vessels. This directly supported the hypothesis that SLIT3 is indispensable for building the necessary neurovascular infrastructure that allows brown fat to function.
To bridge the gap to human relevance, the team extended their investigation to human fat tissue samples from over 1,500 individuals, including those with obesity. Focusing on the gene responsible for producing SLIT3, which previous studies had linked to obesity and insulin resistance, they found that SLIT3 gene expression appears to regulate fat tissue health, inflammation, and insulin sensitivity in obese individuals. "That really got our attention, as it suggests that this pathway could be relevant in human obesity and metabolic health," Dr. Shamsi noted, underscoring the potential translational impact of their work.
The Global Obesity Challenge and Current Treatment Landscape
The urgency for diverse and effective obesity treatments cannot be overstated. Obesity has reached epidemic proportions globally, recognized by the World Health Organization as a major public health crisis.
-
Prevalence and Health Burden: According to the Centers for Disease Control and Prevention (CDC), the prevalence of obesity in the United States alone was 41.9% in 2017-2020. Globally, over 1 billion people are obese. The condition is a leading risk factor for numerous non-communicable diseases, including cardiovascular diseases, diabetes, musculoskeletal disorders, and certain types of cancer. The economic burden is staggering, with direct and indirect costs running into hundreds of billions of dollars annually in the U.S. alone. This immense health and economic toll highlights the critical need for continued innovation in weight management strategies.
-
The Rise of GLP-1s and Their Limitations: The introduction of GLP-1 receptor agonists has been a game-changer for many individuals struggling with weight loss, offering significant reductions in body weight and improvements in metabolic health markers. However, these medications are not universally effective, can have side effects, and often require long-term administration to maintain weight loss. Furthermore, they primarily address the input side of the energy balance equation. A complementary approach that boosts energy output could offer synergistic benefits or serve as a viable alternative for individuals who do not respond well to GLP-1s or prefer a different mechanism of action. The NYU research provides a promising avenue for this complementary strategy.
Paving the Way for Novel Therapies: Harnessing Energy Expenditure
The detailed understanding of the SLIT3-PLXNA1 pathway opens up exciting new possibilities for therapeutic development. Unlike existing drugs that focus on reducing appetite, therapies derived from this research would aim to increase the body’s energy expenditure, essentially turning up its internal thermostat to burn more calories from stored white fat and circulating glucose and lipids.
-
Optimizing, Not Just Growing, Brown Fat: The research suggests that the goal may not necessarily be to grow more brown fat tissue, but rather to optimize the function of existing brown fat. By harnessing the SLIT3-PLXNA1 pathway, scientists envision developing therapies that could "renovate" or "upgrade" an individual’s existing brown fat, making it more efficient at receiving neural signals and drawing in nutrients for thermogenesis. This could involve drugs that mimic the action of SLIT3 fragments, enhance BMP1 activity, or target the PLXNA1 receptor to bolster the neurovascular infrastructure.
-
Potential Therapeutic Applications: The implications extend beyond simple weight loss. By increasing the metabolic activity of brown fat, such therapies could also improve glucose homeostasis and lipid profiles, offering benefits for individuals with type 2 diabetes, insulin resistance, and dyslipidemia. The ability to enhance the body’s natural calorie-burning capacity could represent a significant advance in metabolic medicine, providing a powerful tool in the fight against a spectrum of metabolic diseases.
Future Outlook and Research Directions
While the findings are profoundly promising, the path from laboratory discovery to clinical application is often long and complex. The immediate next steps for Dr. Shamsi’s team and the broader scientific community will involve further preclinical studies to thoroughly evaluate the safety and efficacy of targeting the SLIT3 pathway. This includes:
- Translational Hurdles: Identifying optimal drug candidates that can modulate the SLIT3-PLXNA1 axis effectively and safely in humans.
- Delivery Mechanisms: Determining the best way to administer such therapies – whether through small molecules, biologics, or gene-editing techniques.
- Long-Term Effects: Investigating potential long-term side effects or unintended consequences of consistently enhancing brown fat activity.
- Personalized Medicine: Exploring how individual differences in brown fat mass, activity, and SLIT3 expression might influence treatment responses, potentially leading to personalized therapeutic approaches.
- Combination Therapies: The possibility of combining SLIT3-targeting therapies with existing weight-loss medications, including GLP-1s, to achieve more robust and sustained weight reduction, warrants exploration.
Expert Perspectives and Broader Impact
The scientific community has greeted this discovery with considerable excitement. Researchers in metabolism and endocrinology recognize the SLIT3 pathway as a novel and compelling target. While acknowledging that more research is needed, the findings provide a refreshing new perspective on obesity treatment. The potential to tap into the body’s inherent thermogenic capacity offers a biologically intuitive strategy that complements existing pharmacological approaches.
From a public health standpoint, the development of treatments that enhance energy expenditure could provide a much-needed additional tool in the arsenal against the global obesity epidemic. It offers hope for individuals who have struggled with traditional weight loss methods or current pharmacological options. Moreover, this research underscores the importance of continued investment in fundamental biological research, as breakthroughs often emerge from a deeper understanding of basic physiological processes.
The research was supported by significant funding from institutions including the National Institutes of Health, the G. Harold and Leila Y. Mathers Charitable Foundation, the American Heart Association, and several university-affiliated centers. This collaborative effort, involving multiple research institutions and funding bodies, exemplifies the interdisciplinary nature required to tackle complex health challenges like obesity. The work of Dr. Shamsi and her colleagues at NYU College of Dentistry marks a pivotal moment, opening a new chapter in the quest to harness the body’s own metabolic machinery for better health.








