The integrity of the intestinal barrier, a crucial line of defense for overall health, is increasingly recognized as a significant factor in disease prevention and therapeutic intervention. This complex, multi-layered structure, with its vast surface area—estimated to be over 4,000 square feet, larger than a tennis court—demands a substantial portion of the body’s energy, approximately 40% of its total expenditure, to maintain its function. Emerging scientific evidence points to the disruption of this intestinal barrier as a potential underlying mechanism in the development of a spectrum of health conditions, ranging from autoimmune disorders like celiac disease to chronic inflammatory bowel diseases.
Understanding Intestinal Permeability: The "Leaky Gut" Phenomenon
Intestinal permeability, commonly referred to as "leaky gut," describes a state where the intestinal lining becomes abnormally permeable, allowing substances that would typically be contained within the digestive tract to pass into the bloodstream. This phenomenon is not merely a theoretical concern; researchers have developed methods to assess it. One such method involves the use of inert substances, like blue food coloring, which can be administered and then traced. In individuals with a healthy intestinal barrier, these substances remain confined to the gut. However, in severely ill patients, such as those with sepsis and compromised gut barriers, these markers have been detected in the blood, underscoring the critical role of intestinal integrity in systemic health.
The implications of a compromised intestinal barrier extend beyond acute illness. Disruptions can occur even in seemingly healthy individuals due to common lifestyle factors and readily available medications. This has led to a growing focus on identifying and mitigating these triggers to promote long-term health and potentially prevent the onset of chronic diseases.
Pharmaceutical Triggers: The Impact of Over-the-Counter Medications
A significant area of concern involves common over-the-counter (OTC) medications, particularly Non-Steroidal Anti-Inflammatory Drugs (NSAIDs). Studies have demonstrated that even a single dose of common pain relievers can compromise the intestinal barrier.
Aspirin and Its Gastrointestinal Effects
Research indicates that taking just two standard 325 mg aspirin tablets, or two extra-strength 500 mg tablets, can measurably increase intestinal leakiness. This finding suggests that even individuals without pre-existing gastrointestinal issues should exercise caution when using aspirin, as it can lead to gastrointestinal barrier dysfunction. The mechanism by which aspirin exerts this effect is believed to involve its impact on prostaglandins, which play a role in maintaining the protective mucus layer of the stomach and intestines. By inhibiting prostaglandin synthesis, aspirin can reduce the protective mechanisms of the gut lining.
Furthermore, the notion that buffered aspirin, which combines aspirin with an antacid, might offer greater gastrointestinal protection has been challenged by scientific inquiry. Studies have revealed that both regular aspirin and buffered formulations, such as Bufferin, can induce multiple erosions in the inner lining of the stomach and intestine. Endoscopic examinations of individuals who took aspirin or Bufferin at recommended doses revealed significant redness and erosions in the gastrointestinal tract in a substantial percentage of participants. alarmingly, these detrimental effects can manifest rapidly, with damage observed as early as five minutes after ingestion.
While acetaminophen (commonly known as Tylenol in the United States) is often considered a safer alternative regarding gastrointestinal damage, its use is contingent on liver health. For individuals with pre-existing liver conditions, acetaminophen may pose significant risks. Intriguingly, some common supplements, like vitamin C, have been observed to potentially exacerbate aspirin-induced increases in gut leakiness, suggesting a complex interplay between dietary factors and medication effects.
NSAIDs and Anaphylaxis: A Deeper Connection
The detrimental effects of NSAIDs on the intestinal barrier may also shed light on their association with certain severe allergic reactions. NSAIDs, including aspirin, ibuprofen, and naproxen, have been implicated in a notable percentage of food-induced anaphylaxis cases. Specifically, their use is associated with a significantly higher risk of life-threatening food allergy attacks. This heightened risk is hypothesized to stem from the ability of these drugs to increase intestinal permeability. When the gut lining is compromised, small food particles can potentially "leak" into the bloodstream, where they may trigger an exaggerated immune response in susceptible individuals, leading to anaphylaxis. This highlights a potential pathway through which common medications can influence systemic immune reactivity.
Lifestyle Factors: Exercise and Alcohol’s Influence on Gut Health
Beyond medications, certain lifestyle choices can also impact intestinal barrier function.
Strenuous Exercise and Transient Gut Leakiness
While exercise is widely recognized for its numerous health benefits, extremely strenuous physical activity can, paradoxically, lead to temporary changes in intestinal permeability. An intense workout, such as an hour at 70% of maximum capacity, can divert a significant volume of blood away from internal organs, including the intestines, and towards working muscles. This reduction in blood flow can cause transient injury to the intestinal lining, resulting in mild gut leakiness. This effect can be further exacerbated if athletes concurrently use NSAIDs, a practice that unfortunately remains common. The combination of physiological stress from intense exercise and the gut-disrupting effects of NSAIDs presents a compounded risk to intestinal integrity.
Alcohol Consumption and Gut Permeability
Alcohol consumption has also been identified as a risk factor for adverse reactions, including food allergy attacks, potentially through mechanisms similar to NSAIDs. Alcohol can increase gut leakiness, allowing substances to permeate the intestinal barrier and trigger immune responses. However, research suggests that abstaining from alcohol can lead to the healing and restoration of gut integrity. This implies that moderating or eliminating alcohol intake can be a beneficial strategy for improving gut health.

Dietary Triggers: The Role of Fat and Processed Foods
Dietary patterns play a pivotal role in maintaining or compromising intestinal barrier function. Elevated consumption of certain macronutrients and food types has been directly linked to negative impacts on gut health.
Saturated Fat and the Gut Microbiome
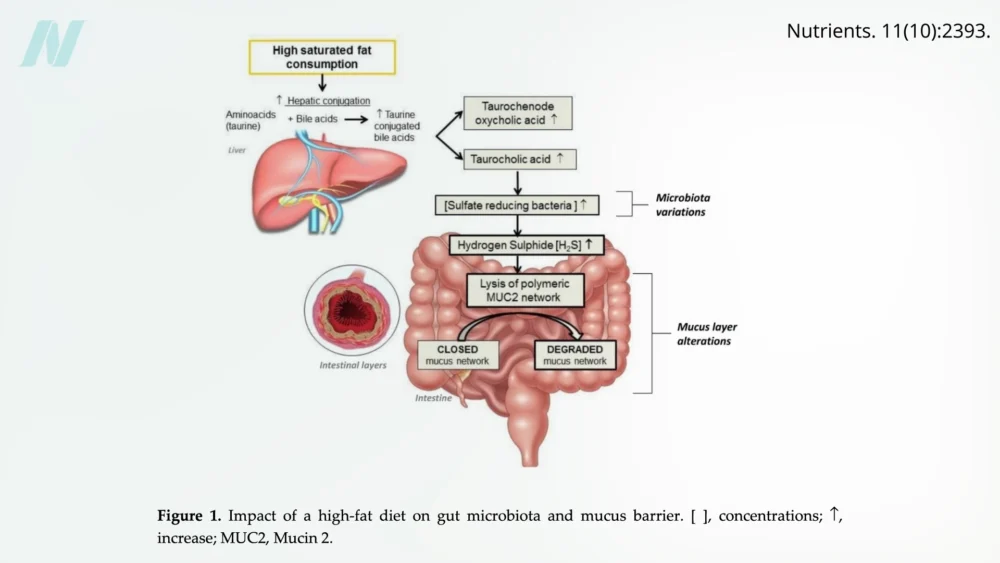
High intake of saturated fats, commonly found in meat, dairy products, and many processed or "junk" foods, has been shown to promote the growth of specific types of bacteria within the gut. These bacteria can produce hydrogen sulfide, a gas with a characteristic rotten-egg smell, which can degrade the protective mucus layer that lines the intestines. This mucus layer is crucial for preventing direct contact between gut contents and the intestinal epithelium, thus maintaining barrier function.
High-Fat Diets and Their Systemic Effects
The broader impact of high-fat diets on intestinal health is a subject of extensive research. While many studies have historically been conducted on animal models or in vitro settings, human interventional trials are increasingly providing valuable insights. There is a strong consensus that high-fat diets, in general, negatively affect intestinal health by disrupting the intestinal barrier through various mechanisms.
The parallel rise in obesity and cardiometabolic disorders with the widespread adoption of Westernized, higher-fat diets has fueled investigations into the causal links. Disturbances in the gut microbiota, the complex ecosystem of microorganisms residing in the intestines, have been consistently associated with an increased risk of these chronic diseases. Rodent studies have indicated that high-fat diets can unbalance the microbiome and impair the gut barrier, leading to disease development.
To bridge the gap between animal studies and human health, comprehensive human trials are essential. A significant six-month randomized controlled-feeding trial provided critical evidence. This study examined the effects of dietary fat on gut microbiota in humans and found that increased fat consumption was indeed associated with unfavorable changes in the gut microbiome and elevated levels of proinflammatory markers in the blood. Importantly, this effect was observed even when refined carbohydrates were replaced with refined fats, such as substituting soybean oil for white rice and wheat flour. These findings underscore the importance of dietary fat intake, even when not primarily saturated, in influencing gut health and systemic inflammation. The implications are significant for public health recommendations, suggesting that countries undergoing dietary transitions should consider advising against increased fat intake, while those already consuming high-fat diets may benefit from strategies to reduce their consumption.
Towards Healing and Prevention: Dietary Strategies
The understanding of factors that disrupt intestinal barrier integrity opens avenues for therapeutic interventions, particularly through dietary modifications. Research into how to heal a "leaky gut" often points towards plant-based diets rich in fiber and antioxidants, which can support a healthy gut microbiome and bolster the intestinal lining.
The Role of Fiber and Whole Foods
Dietary fiber, abundant in fruits, vegetables, whole grains, and legumes, serves as a primary food source for beneficial gut bacteria. These bacteria ferment fiber to produce short-chain fatty acids (SCFAs), such as butyrate, which are essential for nourishing the cells of the colon and maintaining the integrity of the intestinal barrier. A diet lacking in fiber can lead to a less diverse and less resilient gut microbiome, potentially compromising barrier function.
Conversely, diets high in processed foods, refined sugars, and unhealthy fats can promote the growth of less beneficial or even pathogenic bacteria, contributing to inflammation and barrier dysfunction.
Broader Implications and Future Directions
The growing body of evidence linking intestinal permeability to a wide array of health conditions necessitates a shift in how we approach disease prevention and management. Recognizing common triggers, from everyday medications to dietary habits, empowers individuals to make informed choices that can safeguard their gut health.
The concept of the intestinal barrier as a therapeutic target is gaining traction within the scientific and medical communities. Further research is crucial to elucidate the precise mechanisms by which various factors influence gut integrity and to develop targeted interventions. Clinical trials investigating the efficacy of specific dietary patterns, probiotics, prebiotics, and novel therapeutic agents for conditions associated with increased intestinal permeability are vital.
The findings underscore the interconnectedness of diet, lifestyle, medication use, and overall health. A proactive approach that prioritizes the maintenance of a robust intestinal barrier, through mindful choices regarding food, beverages, and medications, holds significant promise for preventing disease and promoting long-term well-being. The ongoing exploration of the gut microbiome and its intricate relationship with the intestinal barrier is likely to yield further insights, paving the way for more personalized and effective health strategies in the future.






