The intricate lining of our intestines, a critical barrier between our internal environment and the outside world, is under constant threat from common everyday substances. Emerging scientific evidence suggests that the integrity of this intestinal barrier, often referred to as a "leaky gut," may represent a significant new frontier in both the prevention and treatment of a wide range of diseases. This complex biological shield, responsible for absorbing nutrients while preventing harmful substances from entering the bloodstream, is a remarkably demanding system to maintain. With its vast surface area, estimated to exceed 4,000 square feet – larger than a tennis court – the intestinal barrier expends approximately 40% of the body’s total energy budget to ensure its proper functioning.
Understanding Intestinal Permeability
Intestinal permeability, a scientific term for the "leakiness" of the gut, is increasingly being recognized as a key factor in health and disease. A healthy intestinal barrier is characterized by tight junctions between epithelial cells, forming a selective gate that allows nutrients to pass while blocking pathogens and undigested food particles. When these junctions are compromised, the barrier becomes more permeable, allowing larger molecules to cross into the bloodstream, triggering inflammatory responses and potentially contributing to a cascade of health issues.
Researchers have employed various methods to assess intestinal permeability. One notable technique involves the use of blue food coloring. In healthy individuals, this harmless dye is expected to remain within the confines of the digestive tract. However, in critically ill patients with severe conditions like sepsis, where the gut barrier is significantly damaged, the blue dye has been detected in their blood, providing a stark visual indicator of compromised intestinal integrity. While severe illness can undeniably lead to a leaky gut, the phenomenon is not exclusive to critical care settings.
Common Culprits: Pharmaceuticals and Everyday Medications
Perhaps one of the most surprising revelations is the extent to which commonly used medications can disrupt this vital barrier. Non-steroidal anti-inflammatory drugs (NSAIDs), a class of drugs widely available over-the-counter for pain relief and inflammation, are frequently implicated. Even a single dose of common pain relievers can have a detrimental effect.
Aspirin’s Impact on Gut Health
Studies have demonstrated that taking just two regular-strength aspirin tablets (325 mg each) or two extra-strength aspirin tablets (500 mg each) can measurably increase gut leakiness. These findings underscore the importance of caution, even for individuals considered healthy, when using aspirin, as it can lead to gastrointestinal barrier dysfunction. This dysfunction can manifest as increased permeability, allowing substances that should remain within the gut to pass into the bloodstream.
The question of whether buffered aspirin, formulated with antacids to theoretically mitigate gastrointestinal irritation, offers greater protection has also been investigated. However, research indicates that buffered aspirin does not significantly differ from regular aspirin in its impact on the gut lining. Studies involving endoscopic examinations have revealed multiple erosions and significant redness in the stomach and intestinal lining of individuals who took either regular aspirin or buffered formulations like Bufferin at recommended doses. Alarmingly, this damage can occur rapidly, with some evidence suggesting it can manifest within just five minutes of ingestion.
Acetaminophen as an Alternative
In contrast to NSAIDs, acetaminophen, commonly known as Tylenol in the United States, may present a safer alternative for pain management concerning gastrointestinal health. While acetaminophen is generally not associated with the same level of gastrointestinal damage as NSAIDs, it is crucial to note its potential risks for individuals with liver conditions.
Vitamin C and Aspirin: An Unexpected Interaction
Intriguingly, some research suggests that vitamin C supplements, often taken for their perceived health benefits, may exacerbate the effects of aspirin on gut leakiness. Rather than offering protective qualities, vitamin C appeared to worsen the aspirin-induced increase in gut permeability in certain studies, highlighting the complex interplay between various substances and intestinal health.
NSAIDs and Food-Induced Anaphylaxis: A Growing Concern
The disruption of intestinal barrier integrity by NSAIDs like aspirin, ibuprofen, and naproxen has been linked to a significant portion of food-induced anaphylaxis cases. These medications are associated with a more than tenfold increased risk of life-threatening allergic reactions to food. The proposed mechanism involves the increased intestinal leakiness caused by these drugs, allowing tiny food particles to enter the bloodstream. When the immune system mistakenly identifies these particles as foreign invaders, it triggers a severe allergic response. This connection raises critical questions about the management of individuals with food allergies and the role of common pain relievers in exacerbating their conditions.
The Demands of Exercise and Alcohol Consumption
Even seemingly healthy activities like strenuous exercise can place temporary stress on the intestinal barrier. An hour of intense physical activity, such as exercising at 70% of maximum capacity, can divert a significant amount of blood away from internal organs, including the intestines, towards the working muscles. This diversion can lead to transient injury and mild gut leakiness. The situation can be further complicated if athletes concurrently use NSAIDs, a practice that is unfortunately not uncommon. The combination of strenuous exercise and NSAID use can potentially aggravate the transient gut permeability, leading to more pronounced effects.

Alcohol consumption also emerges as a risk factor for food allergy attacks, operating through a similar mechanism of increasing gut leakiness. However, the good news is that abstaining from alcohol can allow the gut to begin healing. This suggests that moderating or eliminating alcohol intake can be a beneficial step in restoring intestinal barrier function.
Dietary Influences: Saturated Fat and the Gut Microbiome
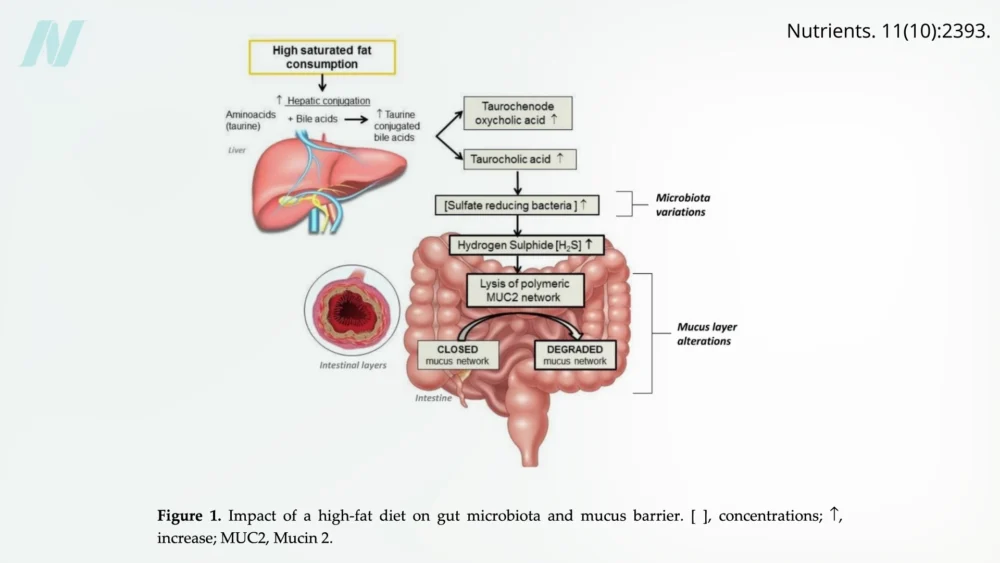
Beyond pharmaceuticals and exercise, dietary choices play a pivotal role in maintaining or compromising intestinal health. Elevated consumption of saturated fats, commonly found in meats, dairy products, and processed "junk" foods, has been identified as a significant contributor to poor gut health.
The Impact of High-Fat Diets
Saturated fats can promote the growth of harmful bacteria within the gut. These bacteria can produce hydrogen sulfide, a gas with a characteristic rotten-egg smell, which can degrade the protective mucus layer lining the intestines. This mucus layer acts as a critical defense, preventing direct contact between gut contents and the epithelial cells. Its erosion leaves the intestinal lining vulnerable.
While much of the research on the negative effects of high-fat diets on intestinal health has been conducted on laboratory animals or in vitro (in petri dishes), human studies are increasingly providing corroborating evidence. The rising rates of obesity and cardiometabolic disorders worldwide have coincided with a shift from traditional lower-fat diets to higher-fat Westernized diets. This epidemiological trend suggests a strong correlation between dietary fat intake and the prevalence of these chronic diseases.
Unbalancing the Microbiome and Impairing the Barrier
A disturbance in the balance of gut flora, the trillions of microorganisms residing in our intestines, has been strongly associated with an increased risk of many of these same diseases. Studies involving rodents have indicated that high-fat diets can disrupt the delicate equilibrium of the microbiome and impair the gut barrier, ultimately contributing to disease development.
To bridge the gap between animal studies and human health, interventional trials are crucial. A six-month randomized controlled feeding trial specifically investigated the effects of dietary fat on the gut microbiota in humans. The findings were significant: higher fat consumption was indeed associated with unfavorable changes in the gut microbiome and elevated levels of proinflammatory factors in the blood. It is important to note that this study did not exclusively focus on saturated fats from sources like meat and dairy. Instead, it involved replacing refined carbohydrates with refined fats, such as substituting white rice and wheat flour with soybean oil. These results carry substantial implications, suggesting that countries undergoing dietary transitions towards Westernized patterns should consider advising against increased dietary fat intake. Conversely, nations that have already adopted such diets may benefit from recommendations to reduce their fat consumption.
Broader Implications and Future Directions
The accumulating evidence on the factors contributing to a "leaky gut" points towards a paradigm shift in how we approach disease prevention and treatment. Understanding and addressing intestinal permeability could unlock new therapeutic strategies for a wide array of conditions, including inflammatory bowel diseases (IBD), celiac disease, autoimmune disorders, and even certain allergies and metabolic syndromes.
The implications extend to public health policy and individual lifestyle choices. Educating the public about the potential gut-disrupting effects of common medications, the importance of a balanced diet, and the role of lifestyle factors like exercise and alcohol consumption is paramount. Further research is needed to elucidate the precise mechanisms by which various substances impact the intestinal barrier and to develop targeted interventions for restoring its integrity.
Addressing Gut Health: A Multifaceted Approach
The journey towards a healthier gut involves a multifaceted approach, encompassing informed choices about diet, medication use, and lifestyle. While the scientific understanding of intestinal permeability is still evolving, the current body of evidence strongly suggests that safeguarding this vital barrier is essential for overall well-being.
Doctor’s Insights on Gut Health
For individuals seeking to deepen their understanding of leaky gut syndrome and explore dietary interventions, further resources are available. Discussions on the inflammatory pathways triggered by animal products and strategies for healing a leaky gut through diet offer valuable guidance. Additionally, the connection between gut health and conditions like Small Intestinal Bacterial Overgrowth (SIBO) highlights the interconnectedness of the digestive system and the importance of a holistic approach to gut health management.
The scientific community continues to explore the intricate relationship between the gut microbiome, intestinal barrier function, and human health. As research progresses, it is likely that novel therapeutic targets and preventative strategies will emerge, further solidifying the central role of a healthy gut in maintaining overall vitality and resilience.






